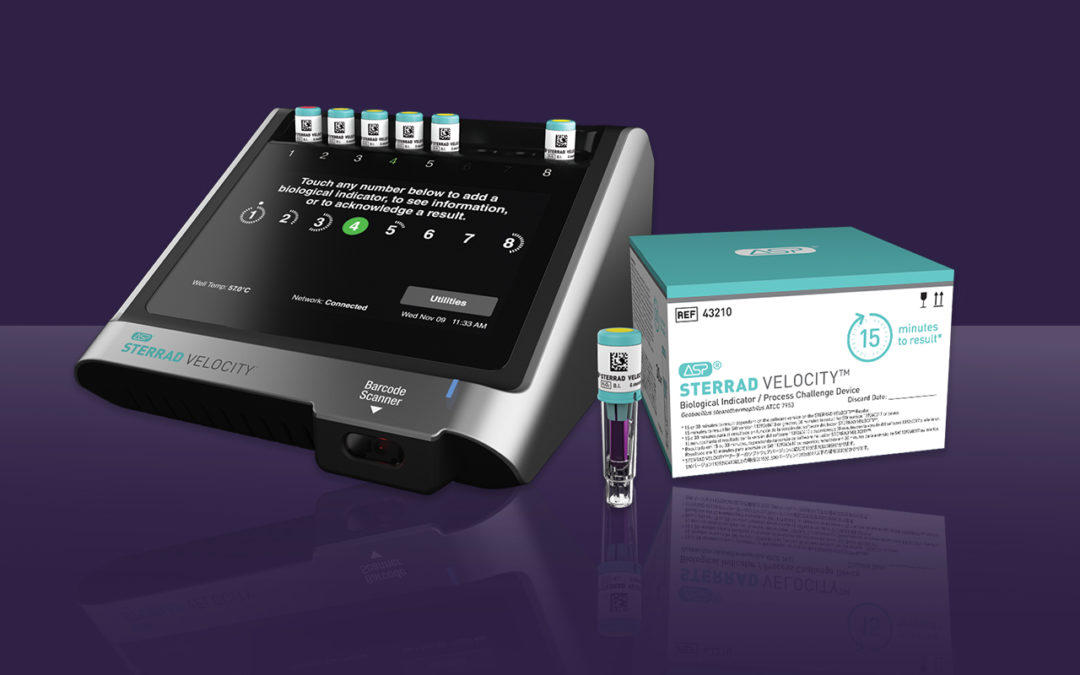
Advanced Sterilization Products (ASP) announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for a 15-minute time to result on the STERRAD VELOCITY® Biological Indicator (BI)/Process Challenge Device (PCD) for use in STERRAD Systems.* The 15-minute time to result is up to 38% faster than competition and offers the fastest way to provide instrument sterility assurance for healthcare professionals. †
“Every minute matters in sterile processing,” notes Amy Smith, VP Global Marketing for ASP. “ASP Innovation to cut the BI read time in half allows customers to assure sterility for every instrument per AAMI guidelines while keeping up with the pace of a busy SPD. This is just another way ASP continues to partner with the health care community to elevate the standard of care by protecting patients at their most critical moments.”
STERRAD VELOCITY BI/PCD is used to monitor loads and assess process performance for vaporized hydrogen peroxide (H2O2) sterilization. This is a crucial step to verify load sterility for critical and semi-critical surgical instruments prior to releasing them to the operating room. AAMI ST58:2013/(R) 2018 states frequent monitoring may mitigate the many factors that could affect cycle variation and reduce risk to patients. It is for this reason AAMI ST58 recommends using a PCD with the appropriate BI daily, but preferably in every sterilization cycle. STERRAD VELOCITY BI/PCD is the only rapid-read PCD validated for STERRAD® Systems that meets AAMI guidelines.
STERRAD VELOCITY BI/PCD is commercially available in the United States. Existing STERRAD VELOCITY System customers should contact ASP to get the 15-minute result with a software upgrade to their reader.
For more information, visit www.ASP.com.
*Fastest BI/PCD currently marketed for STERRAD® Systems. 15 or 30 minutes to result dependent on the software version on the STERRAD VELOCITY® Reader. 15 minutes to result for SW version 1139260410 or greater; 30 minutes to result for SW version 139260317 or below.
† Compared to STERIS Celerity™ 20 HP Biological Indicator and 3M™ Attest™ Rapid Readout Biological lndicator 1295 for Vaporized Hydrogen Peroxide Sterilization
The third party trademarks used herein are the properties of their respective owners.