
Francis Medical Inc., a privately held medical device company, announced the first commercial procedure using the Vanquish Water Vapor Ablation System at Pinnacle Surgery Center in Austin, Texas, by Dr. Eric Giesler with Austin Urology. The procedure was performed on a patient diagnosed with intermediate risk, localized prostate cancer, like those in the company’s VAPOR 2 pivotal study. This first procedure marks a major milestone for the company and the millions of men looking for a minimally invasive alternative to ablate targeted prostate tissue with lower potential risk of the side effects common with traditional therapies.
The Vanquish System is a transurethral, ultrasound and electromagnetically guided, thermal water vapor device used to convectively ablate targeted prostate tissue. The Vanquish System received 510(k) clearance from the FDA in November 2025. This FDA clearance was supported by positive 12-month follow-up data on the first 110 patients enrolled in the company’s ongoing VAPOR 2 clinical study. The VAPOR 2 study is a prospective, multicenter, single-arm study of 235 patients with intermediate-risk, localized prostate cancer across 26 U.S. clinical sites. Performed on an outpatient basis, initial procedures were completed in February 2025, and patients will continue to be followed to gather longer-term outcomes to support a premarket approval (PMA) submission for an expanded indication for the management of clinically localized prostate cancer.
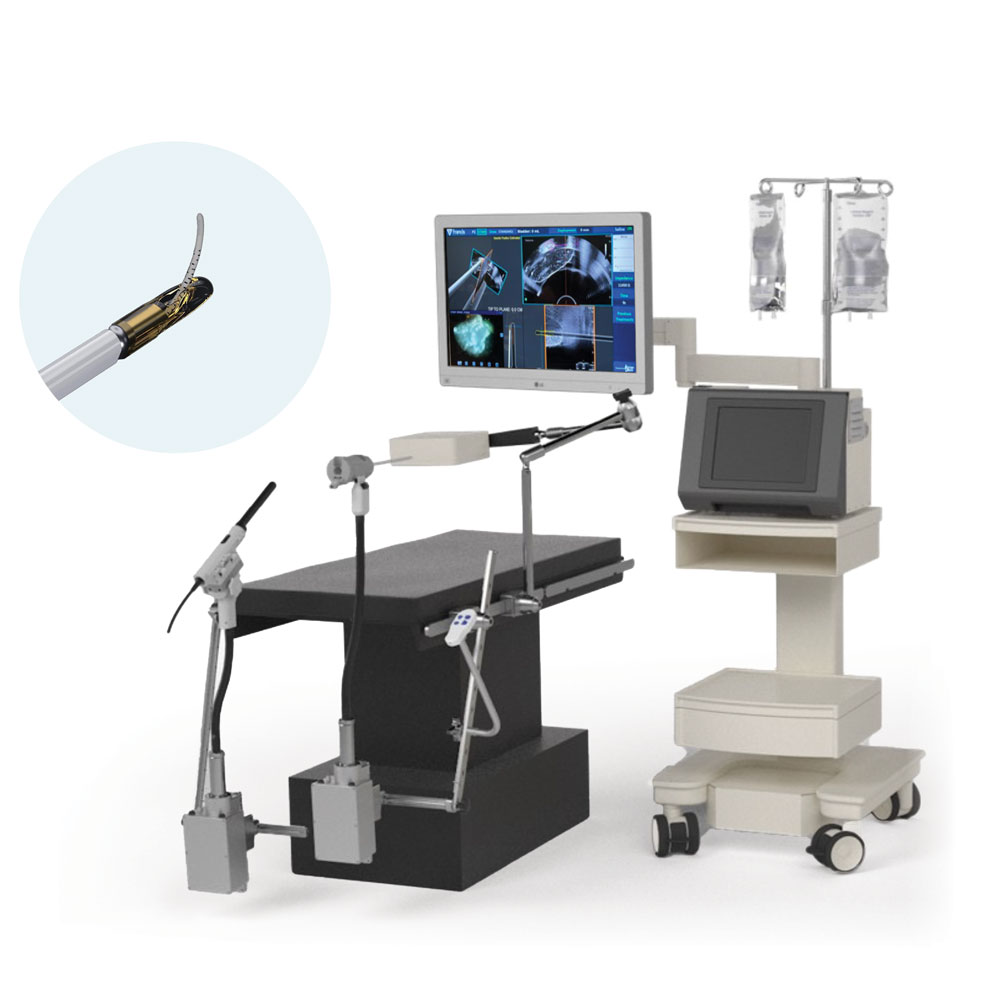
“As a VAPOR 2 clinical study site, it is gratifying to see this innovative technology receive FDA clearance,” said Giesler. “This allows me to offer patients an option that is designed to deliver reliable ablation while potentially reducing the risk of side effects. Our first commercial procedure with the Vanquish System underscores Urology Austin’s commitment to bringing rigorously evaluated, advanced technologies to our community.”
Six-month biopsy data on the first 110 patients in the VAPOR 2 study demonstrated elimination of targeted MRI visible, biopsy confirmed, intermediate risk (≥GGG2) disease in 91% of patients following a single Vanquish procedure. There were no device-related serious adverse events and low rates of urinary incontinence and erectile dysfunction, two common side effects in traditional prostate cancer treatments, such as surgery or radiation. Additionally, the Vanquish System demonstrated the ability to effectively ablate target lesions in all prostate locations, addressing a significant limitation of existing options.
Patient experience and satisfaction results have also been extremely encouraging in the VAPOR 2 study so far. Patients indicated minimal to no pain after the Vanquish procedure and at 12 months post procedure, 93% of patients were extremely satisfied or satisfied, and 94% believed that receiving the treatment was a wise decision.
“Today marks a milestone that took years of innovation, collaboration and clinical partnerships to achieve,” said Dr. Christopher Dixon, Francis Medical chief medical officer. “I am proud of the Vanquish System and the potential this technology has to deliver clinical outcomes without negatively impacting patients’ quality of life, as is commonly seen with traditional treatment options.”
Disclosure: The Vanquish Water Vapor Ablation System has been cleared as a tool for the thermal ablation of targeted prostate tissue via a transurethral approach. The FDA has not evaluated the Vanquish System for the treatment of any disease, including prostate cancer, or evaluated the Vanquish System for any long-term prostate cancer treatment outcomes.










