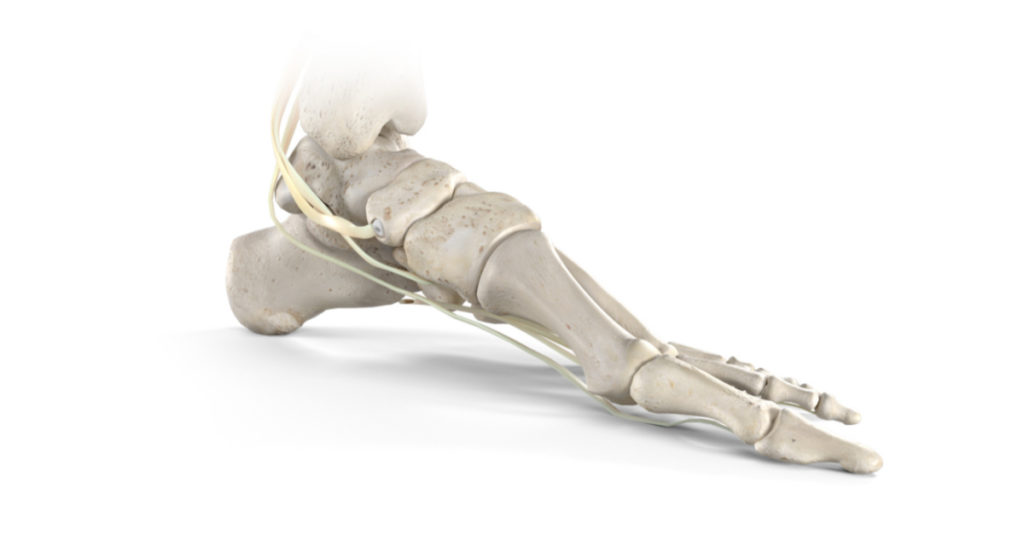
Stryker’s Trauma & Extremities division has launched its Citrelock Tendon Fixation Device System. The new system provides surgeons a differentiated design via a tendon thread featuring a resorbable technology, known as Citregen, that has unique chemical and mechanical properties for orthopaedic surgical applications. Stryker will debut Citrelock at the American Orthopaedic Foot & Ankle Society (AOFAS) Annual Meeting, Sept. 22-25, 2021, in Charlotte, N.C.
“Our customers are looking for a more predictable and effective bioresorbable material than what is currently on the market,” said Michael Rankin, VP Marketing and Medical Education, Stryker Foot & Ankle. “The Citrelock Tendon Fixation Device System helps fill that need with its innovative material technology and unique design. Citregen is an exciting addition to Stryker’s existing biomaterial portfolio and will be expanded for use in additional Stryker Trauma & Extremities indications in the future.”
The Citrelock Tendon Fixation Device System offers surgeons:
- A controlled and homogeneous resorption process that prevents bulk degradation and chronic inflammation.[1]
- Compressive strength that is comparable to cortical bone with a modulus comparable to cancellous bone.[1,2]
- Citregen contains citrate, calcium, and phosphate molecules that are inherent to the bone anatomy.[1]
- Material polymer structure that mimics the extracellular matrix protein network.[1]
- Citregen maintains structural integrity during the healing phase, while the implant is replaced by host tissue over time.[1]
“By leveraging Citregen’s unique material properties, the Citrelock Tendon Fixation Device System introduces design features to ease insertion and secure fixation without damaging the tendon during placement,” said Wayne Berberian, MD, orthopaedic surgeon at HMH Hackensack University Medical Center. “It has been exciting to observe the development of a biomaterial for use in orthopaedic applications. I’m excited to start using Citrelock in my practice.”
Citrelock will be featured during one of Stryker’s industry sessions at AOFAS on September 24. The company is also hosting a new product reception highlighting the benefits of Citrelock along with other recent additions to its Foot & Ankle portfolio. For more information, visit www.stryker-trauma.live/aofas-2021/ or stop by the Stryker booth (No. 425).










