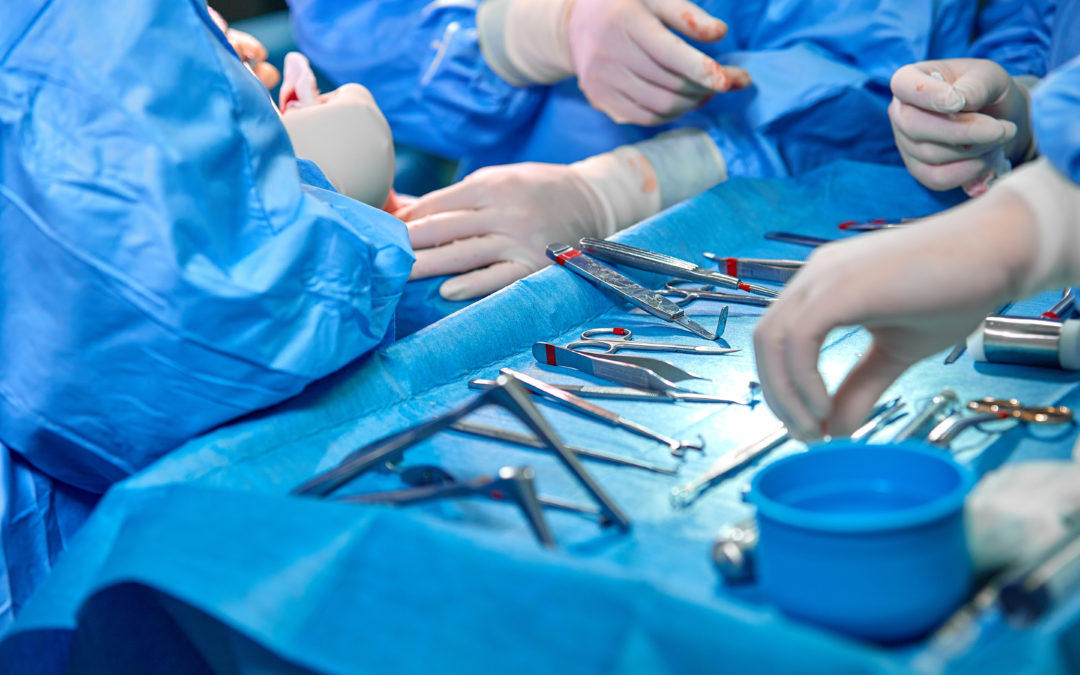
New data published today suggest that personnel who process reusable medical instruments and equipment may be frequently exposed to tissue, blood, and patient fluids even when wearing recommended personal protective equipment (PPE). The findings, which appear in the American Journal of Infection Control (AJIC), the journal of the Association for Professionals in Infection Control and Epidemiology (APIC), detail the results of a pilot project evaluating splash generation during processing activities in equipment decontamination areas.
The first real-world evaluation of PPE effectiveness for sterile processing personnel, the project found that PPE, even when properly donned and doffed, does not provide adequate protection from routine sterile processing activities. The authors highlight the need for more research to support evidence-based guidelines and instructions for safe processing.
“This pilot documented substantial splashing and droplet dispersal during manual cleaning of medical instruments, and personnel got wet even though they were wearing all the recommended PPE,” said Cori L. Ofstead, president and CEO of Ofstead & Associates, and the paper’s lead author. “A combination of engineering solutions such as physical barriers and automated cleaning systems, better protective gear, and splash/exposure-reducing practices is needed to reduce environmental contamination and personnel exposure in sterile processing and endoscopy departments.”
To conduct their research, Ofstead and colleagues affixed moisture-detection paper to environmental surfaces and PPE in the sterile processing department (SPD) of a large, urban hospital. Highly trained, processing personnel then simulated performance of routine reprocessing tasks, including filling a sink, brushing a ureteroscope, and using a power sprayer when rinsing the sink basin. Following completion of the tasks, researchers detected splashes and droplet dispersal of at least three, and in some instances up to five, feet from the sink. This dispersal was associated with all but one of the simulated sterile processing activities. Additionally, currently recommended PPE did not adequately protect SPD personnel from exposure to water and cleaning solutions.
The findings have implications for thousands of SPD personnel at hospitals, clinics, and ambulatory surgery centers, including the nearly 40,000 members of the International Association of Healthcare Central Service Materiel Management (IAHCSMM)—the professional association for certified sterile processing professionals.
“The results from this important real-world evaluation establish the need for additional research by auditors, regulators, and other decision makers to reassess the guidelines for personnel exposure and environmental contamination due to splashes generated in instrument processing areas,” said Ann Marie Pettis, BSN, RN, CIC, FAPIC, and APIC 2021 president. “Until then, controls such as splash-reduction training, correct donning and doffing of PPE, maintaining dirty-to-clean workflow, and cleaning and disinfecting workspaces between activities represent the best approach to reduce cross-contamination and protect sterile processing personnel from exposure to infectious pathogens.”
REFERENCE:
“Droplet dispersal in decontamination areas of instrument reprocessing suites,” by Cori L. Ofstead, Krystina M. Hopkins, Abigail G. Smart, and Marie K. Brewer, was published online in AJIC on December 2, 2021. https://doi.org/10.1016/j.ajic.2021.10.023.