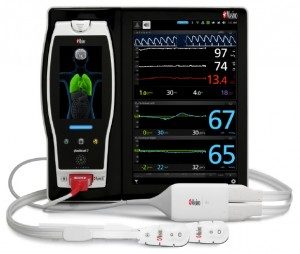
 Masimo has announced the CE marking for the pediatric indication for O3 regional oximetry with the O3 pediatric sensor. Regional oximetry, also referred to as tissue or cerebral oximetry, helps clinicians monitor cerebral oxygenation.
Masimo has announced the CE marking for the pediatric indication for O3 regional oximetry with the O3 pediatric sensor. Regional oximetry, also referred to as tissue or cerebral oximetry, helps clinicians monitor cerebral oxygenation.
O3 regional oximetry uses near-infrared spectroscopy (NIRS) to continuously monitor absolute and trended regional tissue oxygen saturation (rSO2) in the cerebral region. Early detection and correction of imbalances in oxygen delivery to the brain and vital organs are important tools in helping patients avoid postoperative morbidity and adverse outcomes. With the release of the O3 pediatric sensor, O3 regional oximetry monitoring of rSO2 is now available to pediatric patients weighing less than 88 pounds.
Masimo O3 regional oximetry and SedLine brain function monitoring are both available on a single platform, Masimo Root – opening up a path to better understanding of the brain.
O3 regional oximetry for use with adults weighing 88 pounds or greater has received FDA 510(k) clearance. O3 regional oximetry for use with pediatric patients weighing less than 88 pounds has not received FDA 510(k) clearance; the O3 pediatric sensor is not currently for sale in the United States.










