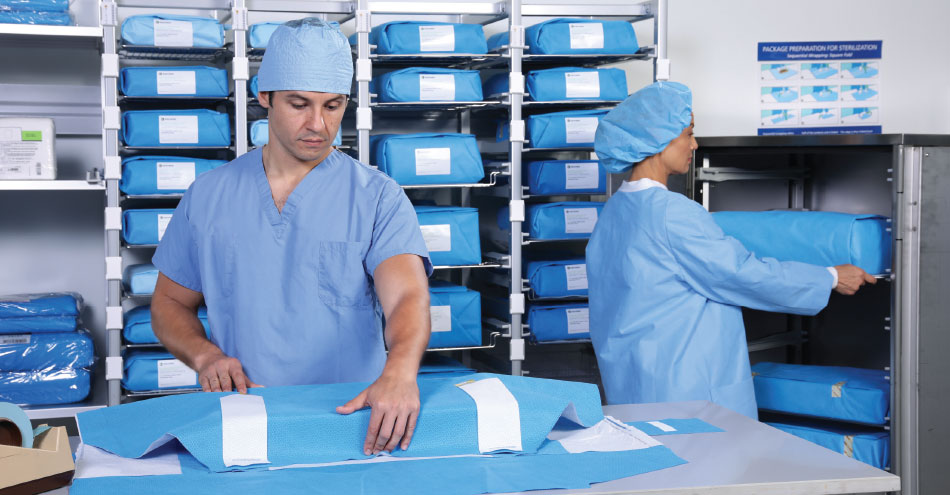
The HALYARD and BELINTRA SMART-FOLD STERISYSTEM is a unique sterilization packaging, transport and storage solution, designed to reduce the risk of tears, cuts and holes in wrapped instrument trays. The system safeguards the sterility of instruments during handling, transport and storage after sterilization until use in the OR. Its stainless steel instrument and transport wire baskets and shelves enhance organization allowing for simpler, faster storage and access to the right instruments at the right time. Additionally, they are FDA approved, have a Medical Class CE certification, and reduce touchpoints to two, exceeding the AAMI standard 79 guideline that designates three.