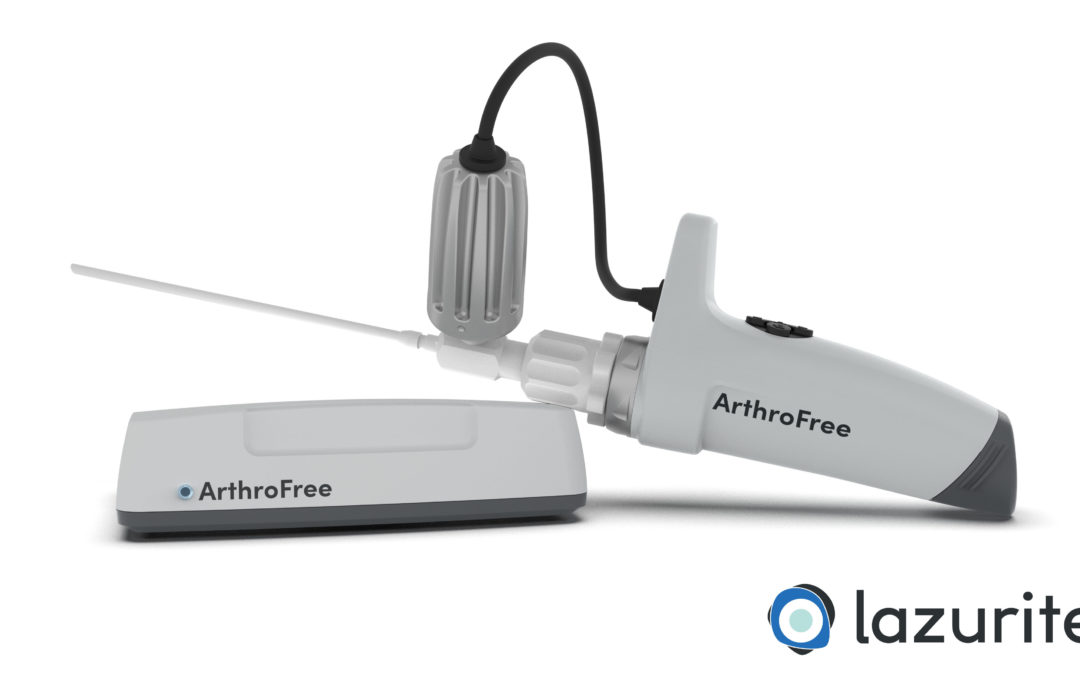
Medical device and technology company Lazurite Holdings LLC has submitted a 510(k) premarket notification to the U.S. Food and Drug Administration (FDA) for its ArthroFree wireless camera system for minimally invasive surgery, and that the submission has been accepted...
READ MORE