Battelle announced that it has begun rapid manufacturing of a system to decontaminate N95 respirator masks and other medical protective equipment.
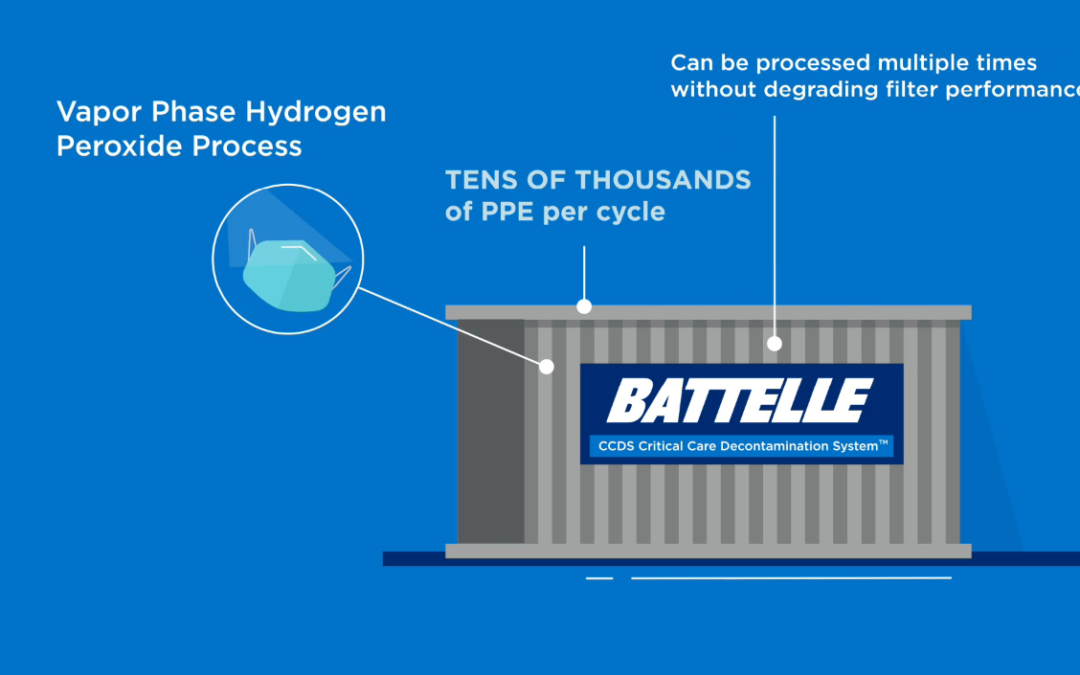
Each Battelle CCDS Critical Care Decontamination System is capable of decontaminating up to 80,000 masks per day at full capacity. Because it is scalable, the system is capable of processing even more pieces of personal protective equipment (PPE) each day. Battelle CCDS uses concentrated, vapor phase hydrogen peroxide (VPHP) and works by exposing used respirator masks to the validated concentration level for 2.5 hours to decontaminate biological contaminates, including SARS-CoV-2.
Battelle has a fully operational system at its West Jefferson, Ohio facility that has processed test batches of previously worn PPE received from Central Ohio hospitals. On March 28, the FDA issued a new Emergency Use Authorization (EUA) to the Battelle Memorial Institute and reissued two EUAs for respirators.
This past week, Battelle started building systems at one of its manufacturing operations in Columbus for shipment to sites around the country. The first completed system is in transit and will be placed at an undisclosed location in the New York metropolitan area to address that city’s critical shortage of PPE needed by health care workers and first responders.
“The Battelle team mobilized to begin acquiring parts and developing manufacturing processes to deploy systems to help those on the frontline who need essential protective equipment,” said Matt Vaughan, Battelle’s Contract Research President. “We are building the next units and are in discussions with health officials across the country to determine where to best place them.”
For decades, Battelle has served the military in protecting troops from chemical and biological hazards. The company operates Biosafety Level 2 and 3 labs where research is performed on live viruses, including SARS-CoV-2. Battelle also has a full medical device development team that works with the FDA on a regular basis.
Battelle CCDS is based on research that Battelle performed for the FDA in 2015 (Richter et al., 2016) to assess the feasibility to decontaminate N95 respirator masks in the event of a PPE shortage resulting from a pandemic. In that FDA study VPHP decontamination, using the same system parameters and critical end points as the current system, was shown to result in 6-log reduction of G. stearothermophilus while not degrading the filter performance of N95 respirators for multiple decontamination cycles. Battelle is currently conducting research to validate that other equipment, including surgical masks and ventilator components, can be decontaminated using this process.
Health care systems that are enrolled in the Battelle CCDS program will collect worn respirator masks daily in accordance with an approved procedure and courier them to one of the active Battelle CCDS Critical Care Decontamination SystemTM locations. The PPE will be labeled with a barcoded serial number for tracking the chain-of-custody throughout the process. This ensures that a hospital system receives its own masks back. Each respirator mask will be marked with the number of times that mask has been processed.
To learn more about Battelle CCDS, please visit www.battelle.org/decon or www.battelle.org/covidnews.