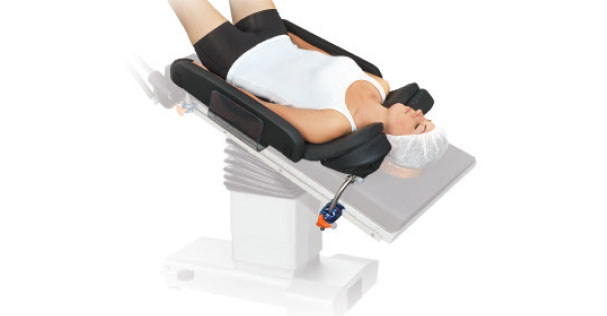
The Alimed SecureFit Trendenlenburg Positioning System(TPS) is a novel, reusable positioning system for Trendelenburg procedures. Developed and tested in conjunction with Massachusetts General Hospital, the SecureFit TPS’ proprietary design combines a uniquely contoured surface geometry to resist sliding with a viscoelastic gel pad to redistribute pressure and protect the patient. No bindings provide unobstructed chest and IV access, and the scapula region is cradled to protect the brachial plexus. The convenient reusable unit offers a simple, consistent, positioning method for laparoscopic, open and robotic procedures, takes less than a minute to set-up and is easy to clean to speed OR turnover.