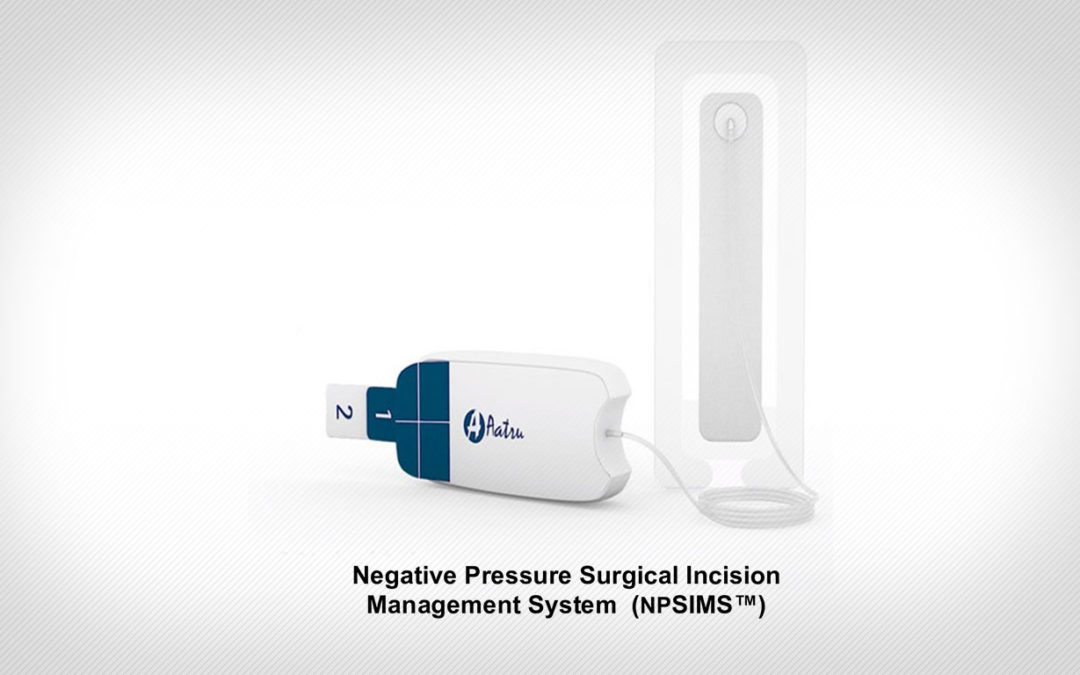
Aatru Medical LLC (Aatru) has announced U.S. Food and Drug Administration (FDA) 510(k) Class II clearance of the NPSIMS Negative Pressure Surgical Incision Management System.
The NPSIMS utilizes an innovative mode-of-action that eliminates the expensive electro-mechanical pump, battery, and electronics found in most other negative pressure wound therapy (NPWT) systems being deployed for closed surgical incision applications. The single-use, disposable NPSIMS is easily activated and operates silently during therapy delivery. A patented preassembled dressing provides ease-of-application with exceptional ‘sealing’ capability while providing patient comfort and ergonometric appeal.
The FDA effort was led by Aatru President and Board member Edward Armstrong who leveraged his extensive professional prior experience with NPWT and medical device companies.
“This innovative NPWT technology allows the NPSIMS product to provide clinical, patient and economic benefits in an elegant presentation, which we look forward to making available to users in the near future,” said Armstrong.
“In spite of the significant challenges posed by the global pandemic, the Aatru team with the support of our regulatory partners successfully received FDA Class II clearance for the NPSIMS. We believe that we are now well-positioned to commercially launch in multiple countries that have expressed immediate interest in our NPWT platform. We have filed more than 60 patents globally to protect our innovative technology, and we have identified manufacturing partners who can immediately scale to meet global demand,” Aatru CEO and Chairman Timothy Wojciechowski said.