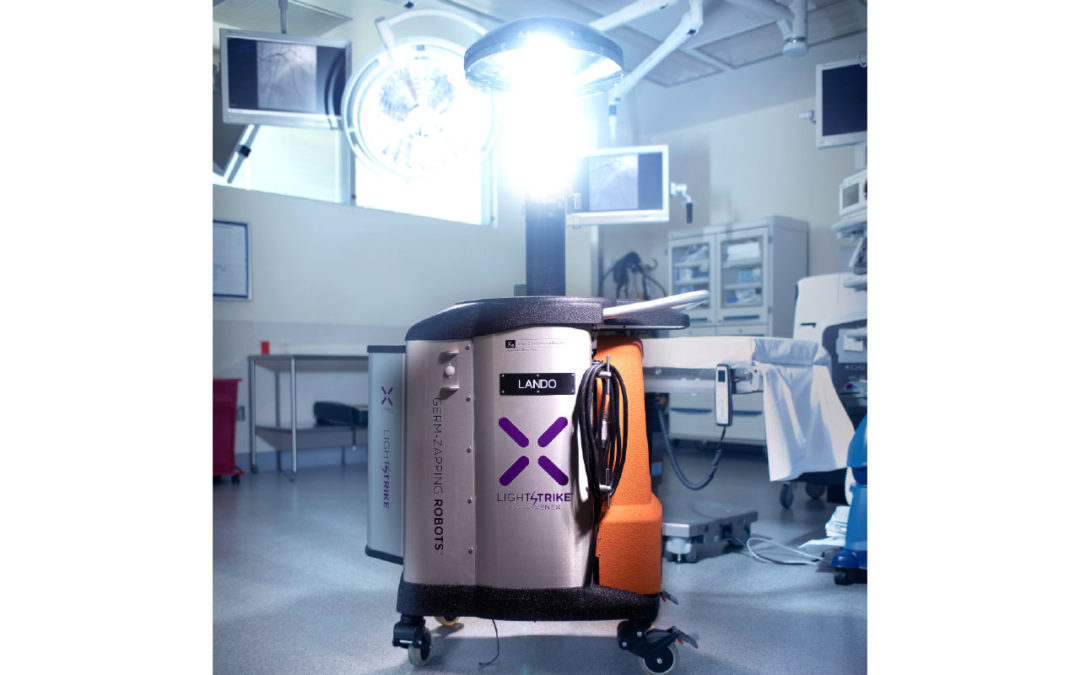
Xenex’s LightStrike Germ-Zapping Robot helps health care facilities reduce SSIs by quickly destroying the pathogens that can cause infections. Hospitals using LightStrike have published peer-reviewed studies showing 46-100% reductions in Class 1 SSI rates in ORs. The only UV room disinfection technology proven effective at destroying SARS-CoV-2 in two minutes (the virus that causes COVID-19), LightStrike robots use a xenon lamp and generate bursts of high intensity, broad spectrum UV light to quickly deactivate viruses, bacteria and spores where they are most vulnerable without damaging expensive OR equipment.