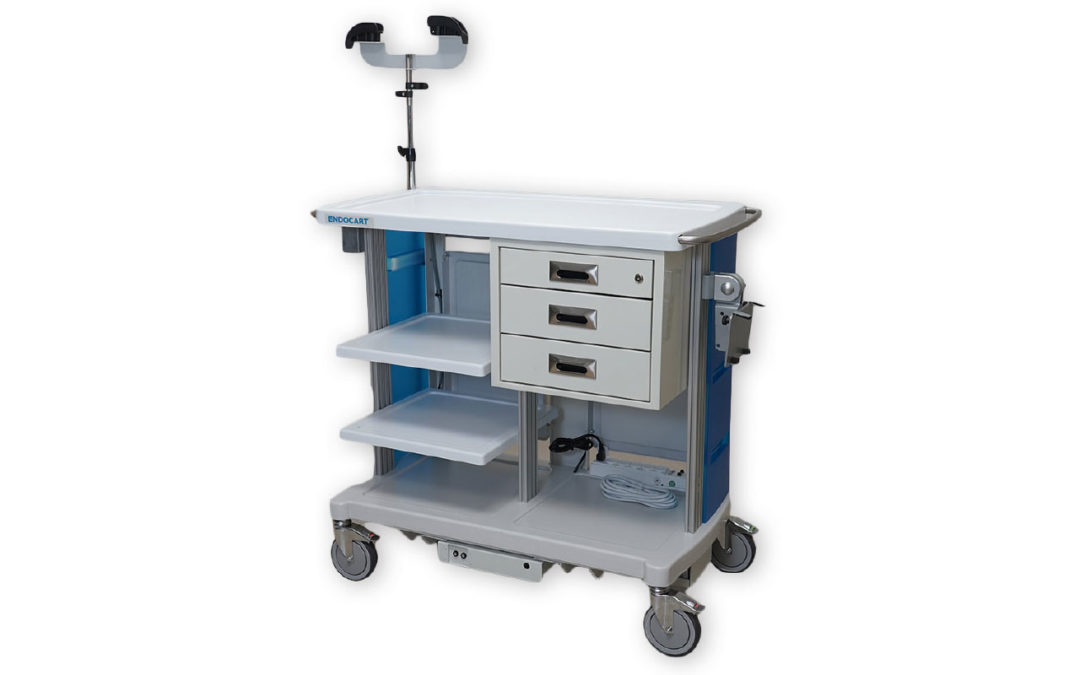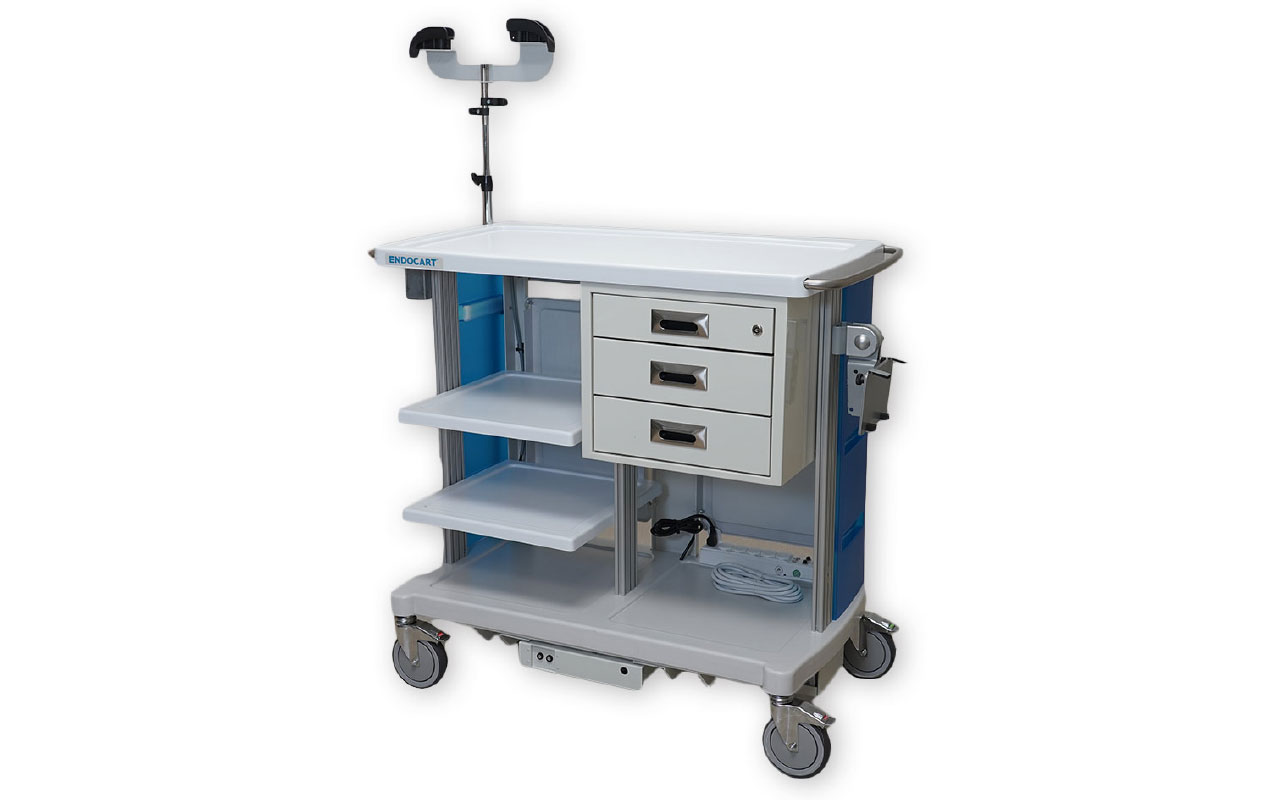
The Endocart was created for GI nurses by GI nurses to help solve the challenges they face every day in the procedure room. Endocart is one of only a few brands of endoscopy carts on the market today. Lightweight and durable, these carts have a variety of customizable options, including monitor mounts, shelves, storage baskets, scope hangers and drawers. The company offers three models including a motorized version for increased efficiency and safety while keeping all medical supplies organized during transport. All models come with an 18-month warranty.
For more information, visit www.endocart.com.