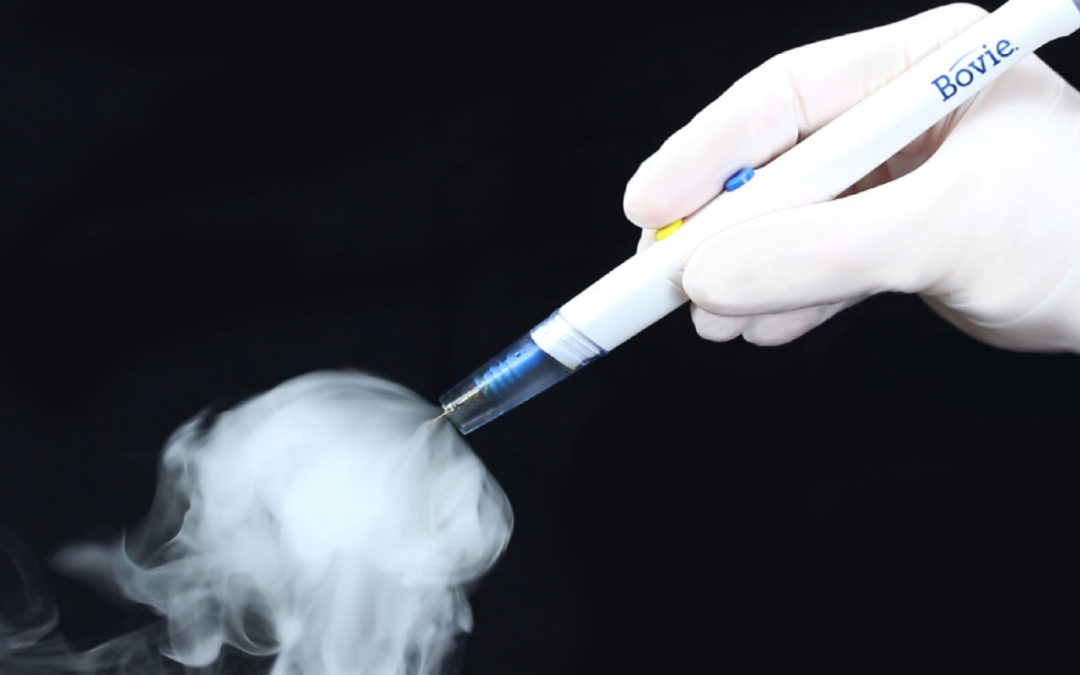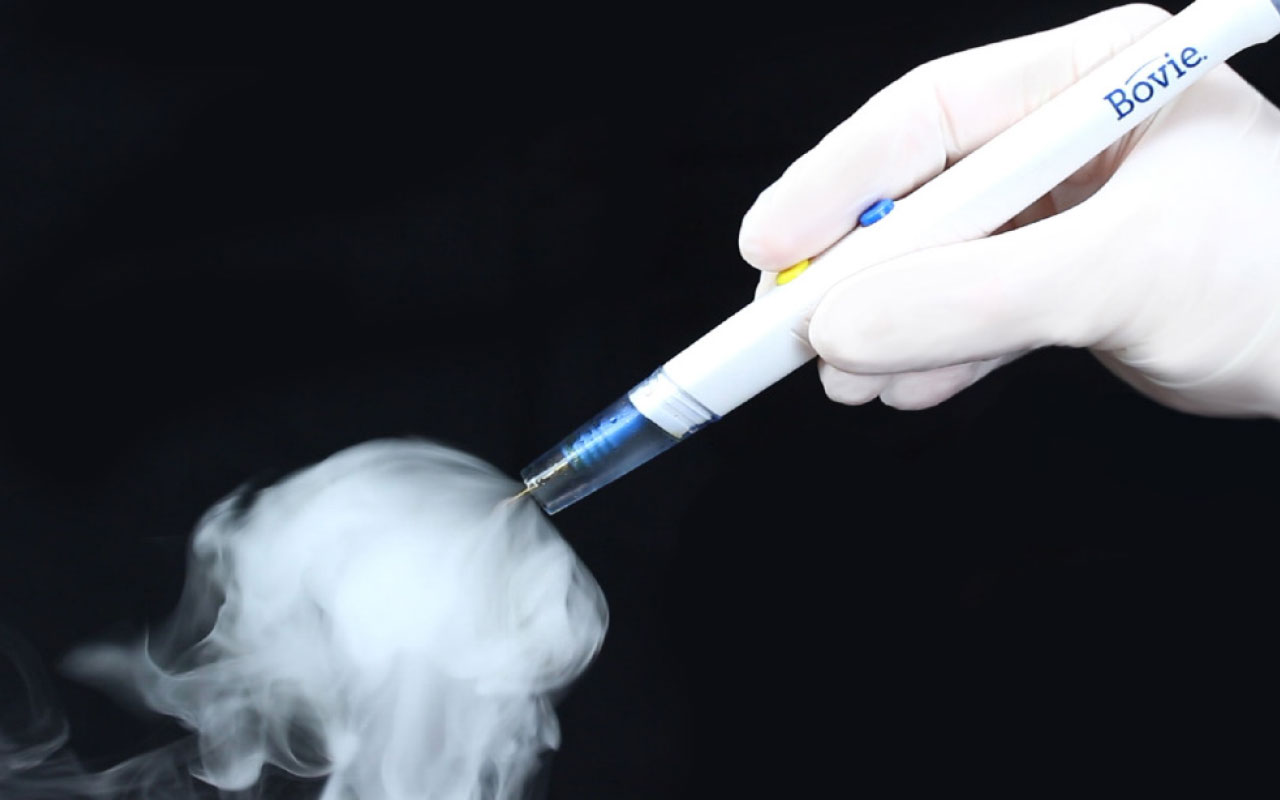
The Orca Smoke Evacuation Pencil is the latest Bovie smoke evacuation solution, a brand synonymous with electrosurgery. The Orca is a powerful, fully integrated, ergonomic, low-profile device providing optimal control and better protection for operating room personnel. The Joint Commission’s – Quick Safety Issue 56 December 2020 lists specific actions organizations should take to help patients and staff reduce the dangers of hazardous surgical smoke plume. Orca provides up to 55% greater smoke plume clearance than the leading competitor at an economic price point. It delivers quality and value combined in one device.