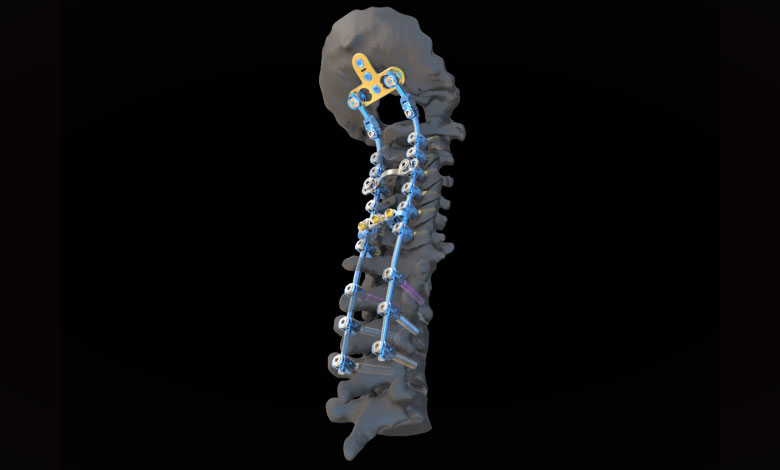
The Johnson & Johnson Medical Devices Companies* has announced that DePuy Synthes has launched the SYMPHONY Occipito-Cervico-Thoracic (OCT) System, expanding its offering for the surgical treatment of conditions in the neck and upper back. The SYMPHONY System includes a differentiated offering of instruments and implants designed for stabilization of the spine in patients undergoing Posterior Cervical Fusion surgery. The announcement was made at the 47th annual Cervical Spine Research Society (CSRS) meeting.
The SYMPHONY System helps streamline procedures and create efficiencies for the surgeon and operating room staff who are treating patients with complex cervical spine disorders. Guided by research conducted in association with the International Spine Study Group, the SYMPHONY System is designed to improve surgeon experience in four key areas: fixation, alignment, targeting and extensions. The range of instrumentation offers options for surgeons to choose from and is compatible with navigation software to aid in targeting in challenging anatomy.
“The most interesting aspect of the SYMPHONY System is its ability to address highly complex needs that we face at the front lines of cervical spine surgery,” said Christopher Ames, MD, Spine Surgeon†. “It also takes into account ways to create efficiencies for healthcare systems, making it a truly comprehensive offering.”
The SYMPHONY System was designed to improve fixation in patients with suboptimal bone quality‡, to address a potential cause of screw failure which may lead to revision surgery. By enabling crossing of the cervical to thoracic junction with one system, the SYMPHONY System reduces the number of instrument trays from six to two§, which may provide cost savings in processing and sterilization as well.1 The system is one of the first posterior cervical systems to be offered with the option of sterile packed implants, which may also reduce processing costs.2
“The SYMPHONY System builds on the deep expertise of DePuy Synthes in treating diseases that affect the cervical spine, and we are excited to elevate our existing portfolio by offering this new enhanced solution for the treatment of these debilitating conditions,” said Nadav Tomer, Worldwide President, Spine, DePuy Synthes¶. “This launch represents an incredible global opportunity to bring a differentiated solution to surgeons looking for reduced complexity, streamlined procedures and more flexibility in treatment options for their patients.”
The system combines the benefits of two market-leading posterior cervical systems—the SYNAPSE OCT System and MOUNTAINEER OCT System—uniting many of the patented features of those brands and building on their proven instrumentation.
*Comprising the surgery, orthopedics, vision and interventional solutions businesses within Johnson & Johnson’s Medical Devices segment
†Consultant to DePuy Synthes Spine
‡The SYMPHONY OCT System is contraindicated for osteoporosis and relatively contraindicated for osteopenia.
§As compared to EXPEDIUM® 5.5mm System and MOUNTAINEER® OCT SPINAL SYSTEM 3.5mm trays.
¶Employee of Medical Device Business Services, Inc.
1. J. Abrams MD, N. Chutkan (2017) Maximizing operating room efficiency in spine surgery: a process of tray consolidation, instrument standardization and cost savings. (#Posete #477) International Society for the Advancement of Spine Surgery.
2. Wells J. The Move to On-Demand Sterile Implants and Instruments: Eliminating Reprocessing Risks.










