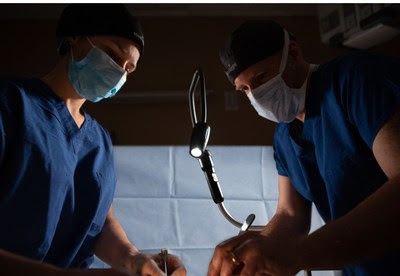
MezLight launches world’s first sterile, flexible, and reusable lighting solution to ease the physical burden on surgeons and enhance lighting in the OR.
MezLight LLC has launched a surgical lighting system to provide surgeons with a bright, sterile and reusable solution for focused light.
Good lighting is an essential element in performing surgical procedures efficiently and accurately to produce the best possible patient outcome. The MezLight was invented by University of Wisconsin transplant surgeon Josh Mezrich in response to his own experience, and that of his colleagues, with the need for better lighting in the operating room (OR).
The current standard for bright, focused lighting is the surgeon’s headlight which has numerous downsides including physical discomfort, encouraging poor ergonomic posture, lack of sterility and contribution to physician distraction during a procedure. Perhaps the most serious is the role headlights make to the already physically demanding activity of surgery. Holding a poor ergonomic posture to keep the headlight trained on the surgical site can ultimately lead to serious physical injury and chronic pain for the surgeon.
MezLight’s surgeon-centric design approach focuses on the wellbeing of the surgeon as a key element in patient outcomes.
“I don’t want to change how people operate; I want to help them operate better,” Mezrich said.
The MezLight attaches to the OR table rail using a standard Clark Socket and delivers the light intensity and illumination performance of a surgeon’s headlight without residing on the surgeon’s head. MezLight’s patent pending flexible neck design allows for use in a wide variety of procedures and provides increased angular cavity illumination not possible with headlights and overhead lights.
Because the MezLight is designed to be sterilized in an autoclave surgeons can control and manipulate their lighting without the need for a towel or sterile sleeve. Interruptions of and distractions to surgical workflow are reduced by the ability of the surgeon to quickly position and reposition the light. The MezLight is FDA registered and available now, details for trialing and ordering a MezLight can be found at www.mezlight.com.









