In the 1960s and 1970s, nursing students were often taught how to manage contaminated objects with their bare hands. This included cleaning patients who were incontinent and counting bloody sponges. In surgery, the scrub nurse would hand off a sponge forceps from the active surgical field, complete with biologic contamination, to the barehanded circulating nurse during sponge counts. Hands often came in contact with microbe-laden material, and the answer was simply handwashing. Nonsterile gloves were not readily available for dirty tasks.
OnCourse Learning guarantees this educational activity is free from bias. The planners and authors have declared no relevant conflicts of interest that relate to this educational activity. See page 43 to learn how to earn CE credit for this module.
The goal of this skin hygiene continuing education program is to provide healthcare professionals with information about the relationship between skin and the transference of pathogenic microorganisms. After studying the information presented here, you will be able to:
- Explain the pros and cons of using gloves as personal protective equipment during patient care
- Describe three natural features of skin that can support or hinder the transference and growth of microorganisms on the caregiver and patient
- Explain how hand hygiene and skin antisepsis work in combination to prevent infection
Historical Background
In 1846, physician Ignaz Semmelweis posited the dangers of microbial transfer from contaminated hands to patients in the obstetric suite of Vienna General Hospital.1 Many postpartum patients of his day died of puerperal fever (e.g., caused by Clostridium sordellii) before he put forth his theories about handwashing and its benefit to safe patient care. Some physicians of his time regarded his theories as nonsense and openly mocked him. Semmelweis noted that more resident physicians’ patients died than did patients of midwives. The key factor was that the residents were not washing their hands after they performed autopsies on women who died from postpartum sepsis and before they attended to other patients’ deliveries.
The use of chlorine during handwashing lowered the maternal mortality rate. Physician Oliver Wendell Holmes Sr. proposed the same theory about handwashing and significantly reduced maternal sepsis and mortality in the United States. Retrospective studies of Semmelweis’ data have shown that handwashing between patient contacts reduced the incidence of infection and lowered the mortality rate.
Gloves and Skin Antisepsis
Today, gloves of all varieties are available in patient care areas for healthcare professionals of all disciplines. Although clinicians wear exam gloves during patient care for self-protection, gloves are not a panacea for dealing with evolving “super bugs” of the 21st century. No glove is 100% impervious. Micropores are present on all glove surfaces.1
The dirty hands of many people reach into the same box of nonsterile exam gloves for personal protection and transmit microorganisms to the box opening and its contents. The opening of the box is exposed to microorganisms on the bare skin of everyone who reaches inside for gloves. As the exam gloves are withdrawn, they come in contact with the contaminated box opening. Microbial contamination can be spread without conscious knowledge.1
The microbial load on any surface is capable of causing postoperative complications, such as infection, wound dehiscence or systemic morbidity. Research reveals that prevention of cross-contamination and surgical site infection consists of appropriate skin antisepsis for both the patient and healthcare professionals across all disciplines. Hand hygiene and skin antisepsis are not singular processes and are effective only during the moment they are performed. Each contact with different areas of the patient’s body and the patient care environment (e.g., bed rails, blood pressure cuff, clothing and stethoscope) requires cleansing of the hands and device, or changing of gloves to prevent deposition of new bacteria. In essence, providers of various disciplines can transfer bacteria from one part of the patient’s body to another because each body part has its own level of bioload. Moving resident flora to another part of the body creates a pathogenic potential for the patient.
Caregivers feel a false sense of security when they don exam gloves and wear them for prolonged periods, performing multiple tasks without removing and changing them. Moisture and heat builds under the gloves, creating favorable living conditions for bacterial reproduction and endospore reactivation. The surface of a glove picks up microorganisms from the environment and deposits them wherever it makes contact. Gloves do not always protect the patient and may, in fact, provide a transfer vehicle for portable pathogens.
Whether at the surgical site or on the hands of the caregiver, skin is laden inherently with resident and transient flora. Inadequate hand hygiene allows opportunistic pathogens in varying life stages to transfer between patients and other surfaces during everyday activities. Studies monitoring handwashing reveal that many people do not wash their hands properly after using the toilet or diapering a baby. This manifests in the OR, where body substances abound, and the risk for transference is a serious concern. Hand hygiene and skin antisepsis in surgery depend on using products according to the manufacturer’s recommendations. The products must be used correctly and provide microbial kill to be effective. Another consideration is the safety of the product.
Reclassification of Antiseptic Solutions
The Food and Drug Administration (FDA) is in process of reclassifying antiseptic solutions used in healthcare as personnel hand washes, personnel hand rubs, surgical hand rubs, surgical hand scrubs and preoperative skin preparations for patients.2 Antiseptic solutions are classified as generally accepted as safe (GRAS) and/or generally accepted as effective (GRAE). Consideration is given to whether the product is applied and remains on the skin (“leave on”) or if the product is rinsed off with water. Absorption through the skin is a concern. Studies have shown that systemic exposure is greater than previously thought with prolonged use. Surgical personnel may be at risk when using the “leave on” products daily during a period of six months.2
Recent studies have shown that prolonged exposure may pose risk to those who are exposed continually to the chemical ingredients contained in antiseptic solutions. In preparation for generating the final rule, research has shown the presence of the chemicals in the blood, urine and tissues of the users. The classifications will also encompass developmental and reproductive toxicity (DART), hormonal effects, toxicity and carcinogenic potential, and progressive antimicrobial resistance. The ability to isolate the chemicals at previously undetected lower levels has raised concern. The chemical triclosan is a common ingredient in consumer and healthcare products. It has a hormonal effect that has been isolated in urine, cord blood and breast milk. Animal studies show thyroid and reproductive changes (estrogenic and androgenic) in newborn and young test mammalian subjects. It is unknown if a maternal exchange has taken place or if an actual DNA gene alteration has occurred.2 Triclosan, originally categorized as a pesticide, is found commonly in products such as fabrics, cosmetics, toothpaste, many antiseptic soaps, toys and sutures. Measurable elevated blood alcohol levels have been found in surgical personnel, who use alcohol-based surgical hand rubs. Advanced technology and newer testing methods have raised many questions that surpass general safety and effectiveness established by the FDA in 1994 when the original antiseptic rule was adopted.2
Contemporary Issues
The risk of death from multisystem organ failure after surgery is doubled if the patient becomes septic after surgery. Skin antisepsis is one way to minimize the risk of infection at the surgical site, but it must be paired with adequate hand hygiene of the surgical team to be effective. Members of the healthcare professional team must collaborate to provide hand hygiene and skin antisepsis, the primary steps in preventing surgical-site infections in all surgical procedures.
In 2008, Medicare started to deny reimbursement to surgical facilities for preventable mediastinitis resulting from contamination during cardiac surgery. Surgical technique and the entire team’s maintenance of a sterile environment during procedures are major factors in preventing infection. Facilities have addressed some of the issues associated with microbial transfer by the healthcare teams by the installation of motion-sensor hand gel dispensers and no-touch paper towel dispensers.
 Physiologic Factors in Asepsis
Physiologic Factors in Asepsis
The skin is a protective barrier that, when intact, minimizes the host’s exposure to UV rays of the sun and prevents absorption of certain toxins, chemicals and penetration by microorganisms. It serves as a thermoregulatory guardian and sensory organ. Anatomically, all the functions of the skin have a synergistic role in the wellness of the body. But some of the functional structures designed to protect the host from infection can create opportunistic avenues for microorganisms to enter the body surreptitiously.
Let’s explore three physiologic protective factors associated with human skin that can be problematic. First is the “intactness” factor of the skin as a whole. The surface of healthy intact skin does not provide favorable living conditions for the resident bacteria on the skin surface.
Balance in the numbers of resident bacteria helps prevent transient bacteria from accumulating. The normal epidermal surface is somewhat dry, salty and avascular with a low pH. This is not a friendly surface for bacterial colonization. But the skin as a physical barrier is easily breached. Small tears or perforations in the skin leave the host vulnerable to microbial entry, thereby changing the survivability factor in favor of transient bacterial growth. Examples of simple breaks include body piercings; hangnails; pimples; and tiny injuries, such as paper cuts. The intentional incising of the patient’s skin can provide a portal of entry for any living microorganism (e.g., methicillin-resistant Staphylococcus aureus [MRSA] or vancomycin-resistant Enterococci) or dormant bacterial endospore (e.g., Bacillus or Clostridium classes).
Second, consider the structure of the skin and its physiologic appendages. Most skin surfaces have hair-bearing follicles, which include the ducts of sebaceous holocrine glands (i.e., oil glands). The face, chest and scalp have the highest number of holocrine glands. The sebum, or oil contained in these glands, is not sterile and contains amino acids and lipids that are nutritious for microorganisms. The follicle provides a locus for microbial growth and transfer across the surface of the hair as it exits the skin surface. Obstruction of oil glands and sequestering of colonizing microorganisms can cause inflammation and an abscess.
Two types of sudoriferous (i.e., sweat) glands, apocrine and eccrine, accompany sebaceous glands in different body locations and offer additional nutritional media and moisture for the growth of S. aureus and Corynebacterium. (Staph grows well in the skin environment and can become resistant.) Release of sweat from apocrine glands in the armpits and groin and from eccrine glands over the remainder of the body surface is part of the thermoregulatory process of the skin. Odors produced in the hair-bearing areas of the body are the result of bacterial growth and degradation. Microbial growth can be augmented by the use of skin-lubricating lotions that can inactivate many antiseptic skin products.
The third consideration is the flexibility and mobility of the skin’s surface. Generally, skin is pliable. Manipulation of the oil and sweat glands increases discharges onto the surface of the skin. A susceptible recipient can be contaminated by pathogens transferred by these body substances if the skin is not intact. Surface cells desquamate daily, and 10% of shed cells carry viable bacteria. Dry, cracked skin sheds more epithelial cells, decreasing microbial growth inhibiters. Increased numbers of living microorganisms or bacterial endospores are transferred to patients or objects in the environment.
Antiseptic Considerations
Ineffective handwashing may be part of the problem. Further investigation is warranted concerning the use of antiseptic soaps in all settings. Overuse of antiseptic soaps, especially at home, may be causing increased resistance in certain bacteria. Products such as dishwashing detergent, deodorant, shampoo and toothpaste used in the home contain 0.15% to 0.3% triclosan as a bactericide. Triclosan blocks lipid synthesis in E. coli by inhibiting the hormonal activity of certain enzymes. Studies have shown that continued use of antiseptic products has prompted the proliferation of drug- and antiseptic-resistant strains of microorganisms in the natural environment. The targeted bacteria are not killed because of inadequate contact time with the bactericide; they continue to reproduce, genetically transmitting resistant plasmids — bacterial DNA molecules capable of self-replication — to the next generation. Pseudomonas aeruginosa has been shown to develop resistance to antibiotics, such as ciprofloxacin (Cipro), after exposure to triclosan.2 Not all bacteria respond in kind, but some experts suggest that triclosan and other antimicrobials are overused in household products and may result in antibiotic and antiseptic resistance in the natural environment.
Skin damaged by repeated washing loses the anatomic flexibility and surface protection of the natural epidermis. Washing damaged skin is not as effective as washing healthy skin.3 As skin health declines, resistant microorganisms colonize the surface. Handwashing increases skin damage and can augment transference of bacteria during patient care. One researcher reports that postscrubbing irritation persisted for several days and the skin did not return to a healthy state for 17 days. Decreased numbers of natural flora on the hands of long-term antiseptic users is followed by decreased resistance to topical infections. Loss of resident flora indicates a shift in skin pH and decline in natural barrier protection. Damaged skin maintains and sheds higher numbers of bacteria. (The pH of intact skin is not good for many skin microbes to overpopulate.)
In Surgery
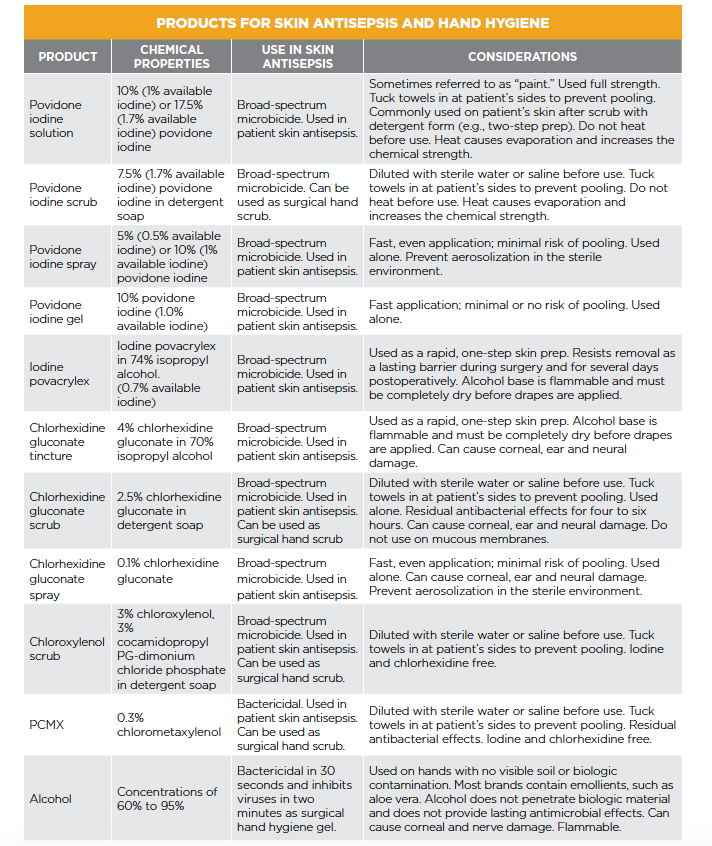
 Many healthcare workers across various disciplines continue to have poor hand hygiene despite best-practice evidence about microbial transfer between individuals. The GRAS/GRAE status of all antiseptic solutions is under investigation, and the results of studies often yield conflicting results.2 (See table on previous page)
Many healthcare workers across various disciplines continue to have poor hand hygiene despite best-practice evidence about microbial transfer between individuals. The GRAS/GRAE status of all antiseptic solutions is under investigation, and the results of studies often yield conflicting results.2 (See table on previous page)
Patient adherence to presurgical showers with antiseptics is inconsistent. One facility had patients bathe upon arrival in the preoperative holding area using prewarmed no-rinse wipes impregnated with 2% chlorhexidine gluconate antiseptic. The usual concentration for the chlorhexidine gluconate surgical soap is 4%. Because it remained in contact with the skin until the initial skin incision, the lower concentration of the chlorhexidine gluconate was effective at reducing bacterial counts, particularly in the hair-bearing regions of the groin and axillae. (In addition, minimal handling of the skin around hair shafts, such as clipping, decreases the risk of infection.) Although no other products were compared, the facility reported a 66% reduction in surgical site infections attributed to use of the chlorhexidine gluconate during the pilot study. A review of preoperative bathing or showering with 4% chlorhexidine gluconate did not show any benefit over cleaning the skin with any antiseptic, even common household soap. The 2% chlorhexidine gluconate no-rinse wipe may have been beneficial because of the mild mechanical exfoliation associated with applying the wipe to the skin and the chemical residue absorbed by epidermal cells. Prewarming the chlorhexidine gluconate cloth to body temperature contributed to patient comfort and adherence.
As far as patients’ skin antisepsis, healthcare professionals as a team should consider the duration of chemical contact, the use of adherent antimicrobial drapes (incise sheets) and one-step preps. Plastic adherent drapes with iodophor or other antimicrobial properties minimize microbial transfer during the surgical procedure, but may cause breakdown of the protective epidermal surface when they are removed at the end of the case. Iodophor (also safe and effective for neuro preps) and alcohol-based one-step preps are applied to the skin and are intended to remain on the patient’s skin for several days postoperatively. Natural epidermal shedding of the skin is supported by the antiseptic coating. Chlorhexidine gluconate is known to bind with the stratum corneum of the epithelium, providing six hours of antimicrobial effect. Attempts to rub one-step prep material off the skin prematurely will cause the loss of natural epidermal protective activity. If removal is necessary, an alcohol-based removal solution is commercially available from the manufacturer.
The Surgical Team
Cognitive variables ranging from one’s personality to attitudes and beliefs influence a person’s practice of hand hygiene and should be discussed collaboratively among all disciplines. Healthcare professionals should perform hand hygiene in different ways using different products according to the situation.4 Antiseptic soap and warm tap water should be used when arriving on duty, before meals, for removing visible soil and before leaving the surgical suite for home. Gel rubs with an alcohol base are used when hands are not visibly soiled or when patient skin or equipment has been handled.5 Gel rubs can irritate the skin and are flammable; however, many gel rubs contain emollients to minimize skin breakdown. The gel rub remains in contact with the skin, offering prolonged chemical action against microorganisms similar to the no-rinse chlorhexidine gluconate wipes.5
 Surgical hand scrubs before gowning and gloving include antiseptic detergent and a sponge-brush apparatus. The sponge-brush mechanically removes skin detris. The detergent component decreases the surface tension of the skin as the antiseptic properties chemically destroy bacteria. The sponge surface causes less skin irritation than the brush portion of the sponge-brush apparatus and is recommended for use on the thinner parts of the hands and arms. Surgical hand and arm scrubbing should include one of the following methods: counted stroke scrub, anatomic scrub by area or timed scrub. Healthcare professionals should clean their fingernails using a nail pick and antiseptic soap under running water. Artificial nails harbor microorganisms and are not permitted in surgical hygiene. Nail polish that is unchipped and less than four days old is acceptable.4
Surgical hand scrubs before gowning and gloving include antiseptic detergent and a sponge-brush apparatus. The sponge-brush mechanically removes skin detris. The detergent component decreases the surface tension of the skin as the antiseptic properties chemically destroy bacteria. The sponge surface causes less skin irritation than the brush portion of the sponge-brush apparatus and is recommended for use on the thinner parts of the hands and arms. Surgical hand and arm scrubbing should include one of the following methods: counted stroke scrub, anatomic scrub by area or timed scrub. Healthcare professionals should clean their fingernails using a nail pick and antiseptic soap under running water. Artificial nails harbor microorganisms and are not permitted in surgical hygiene. Nail polish that is unchipped and less than four days old is acceptable.4
Gel antiseptic rubs are appropriate for use after removing surgical gloves. However, powdered gloves can leave residue on the hands that cause irritation, and the residue should be washed off with soap and water. Gel antiseptic rubs do not remove debris or soil; therefore, hand washing with an antiseptic product is necessary for adequate hand hygiene.6 Skin is never rendered sterile, but controlling microbial load at an irreducible minimum is important for both the patient and caregiver.
Nonsterile members of the surgical team wear gloves during routine patient care as a component of personal protective equipment. Sterile gloves are used commonly in place of nonsterile exam gloves because they conform to the contours of the hand and feel like a second skin for intubating and suctioning patients. Unfortunately, many nonsterile members of the anesthesia team wear the same pair of gloves throughout the entire surgical case after being exposed to patient body substances during intubation. Gloves can become comfortable and feel natural to the wearer. For example, some anesthesia personnel adjust monitors and press keys on a computer keyboard wearing the same gloves used for oropharyngeal procedures and then inject medications, such as propofol (Diprivan), into the patient’s IV.2 The same computer keyboard is used or monitor button pressed several times during the day by many anesthesia personnel who, in turn, render patient care with or without gloves. Transference of living microorganisms or bacterial endospores between surfaces in the OR and the patient is a demonstrated reality.
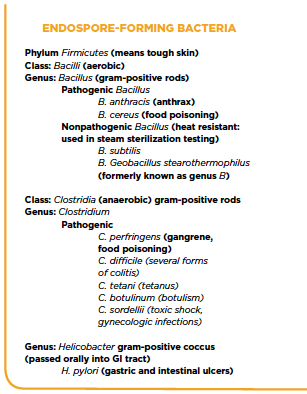
Any residual microbial contamination can be transferred from the inanimate surface to the patient and possibly to injectable medication. Studies have shown that certain microbes can persist on surfaces more than 24 hours and can be transferred from gloved or nongloved hands to multiple surfaces. Dried bacteria can form resistant endospores as self-protection and later reanimate and replicate themselves by binary fission when conditions again become favorable, such as entry into a host. Examples of endospore-forming bacteria of particular concern include several genera of Bacillus and Clostridium (e.g., Bacillus cereus, Clostridium difficile and Clostridium perfringens). (See examples of endospore-forming bacteria on the previous page.)
Examples of Endospore-Forming Bacteria
Endospore-forming bacteria are gram-stain positive. Endospores are formed in response to environmental changes on skin and inanimate surfaces that do not support active bacterial growth and reproduction. The process takes about eight hours and is known as sporulation. Endospores are protective capsules that form inside bacteria to resist ultraviolet and gamma radiation, temperature changes, desiccation and many chemicals in the environment; they cause the outer covering of the bacterial cell to shed. When living conditions are favorable, the endospore reanimates and reproduces by binary fission because the genetic material of the bacterium has been preserved.
Gloved hands carry a larger microbial load because of the nature of the glove material’s texture. The most common bacteria isolated from keyboards were coagulase-negative staphylococci, bacillus and MRSA. Best practices indicate that gloves should be removed after any procedure, and hands should be washed or treated with an alcohol-based gel rub.6 Healthcare professionals as a team should perform hand hygiene several times during patient care if they are handling multiple areas of the patient’s body or touching multiple items throughout the patient care environment. Gloves are never 100% impervious, and the wearer could be contaminated during patient contact. A study in Australia testing the porosity of surgical gloves proved that all gloves tested were porous enough to transfer bacteria from the wearer’s hands to the patient and vice versa.
Proper skin antisepsis and hand hygiene can minimize surgical site infections, and healthcare professionals across disciplines should collaborate to enhance adherence. Skin antisepsis includes surgical techniques, such as the use of antimicrobial adherent incise drapes and antiseptic solutions that bind with skin cells.2,4 Hand hygiene benefits the member of the surgical team as well as the patient. Knowing which product and method to use is essential to preventing surgical site infections. Given the microporosity of surgical gloves, healthcare professionals as a team must use skin antiseptic products appropriately so they will provide an effective microbial kill and minimize microbial transfer between wearer and patient.
Nancymarie Phillips, PhD, RN, RNFA, CNOR, is professor of perioperative education at Lakeland Community College, Kirtland, Ohio.
References
1. Phillips NM. Berry and Kohn’s Operating Room Technique. 12th ed. St. Louis, MO: Elsevier; 2012.
2. Safety and effectiveness of health care antiseptics: topical antimicrobial drug products for over-the-counter human use; proposed amendment of the tentative final monograph; reopening of administrative record. A proposed rule by the Food and Drug Administration on 05/01/2015. Federal Register Web site. https://www.federalregister.gov/articles/2015/05/01/2015-10174/safety-and-effectiveness-of-health-care-antiseptics-topical-antimicrobial-drug-products-for#h-52. Published May 1, 2015. Accessed May 29, 2015.
3. Guidelines for Perioperative Practice. Denver, CO: The Association of PeriOperative Registered Nurses. 2015.
4. Spruce L. Back to basics: hand hygiene and surgical antisepsis. AORN J. 2013;98(5):449-457. doi: 10.1016/j.aorn.2013.08.017.
5. Howard JD, Jowett C, Faoagali J, McKenzie B. New method for assessing hand disinfection shows that preoperative alcohol/chlorhexidine rub is as effective as a traditional scrub. J Hosp Infect. 2014;88(2):78-83. doi: 10.1016/j.jhin.2014.06.013.
6. Macinga DR, Edmonds SL, Campbell E, McCormack RR. Comparative efficacy of alcohol-based surgical scrubs: the importance of formulation. AORN J. 2014;100(6):641-650. doi: 10.1016/j.aorn.2014.03.013.
Clinical Vignette
Hannah was admitted to a community hospital for treatment of increased intracranial pressure caused by a tumor that obstructed the flow of cerebral spinal fluid. She was taken to the OR for placement of a ventriculostomy for external drainage of the cerebral spinal fluid. In the OR, the hair around the portal site was clipped. Her remaining hair was secured away from the sterile field. A two-step iodophor prep was performed. Chlorhexidine gluconate is neurotoxic and was not used.
The sterile team scrubbed with iodophor and used sterile technique for gowning and gloving. They used an iodophor incise sheet as part of the draping procedure. Irrigation was done with room temperature sterile lactated Ringer’s solution. Antibiotic prophylaxis was not used before, during or after the surgical procedure. The cerebral spinal fluid was collected for baseline culture and sensitivity; it was negative.
On postoperative day two, Hannah’s temperature was 102 F, and the cerebral spinal fluid appeared slightly cloudy. The cerebral spinal fluid cultures grew Staphylococcus aureus. During her morning care, the nurses aides had manipulated the three-way stopcock attached to her ventriculostomy port without first using adequate hand hygiene, causing an ascending catheter-associated infection. Hannah received antibiotics intrathecally, and her cerebral spinal fluid cultures resolved. By postoperative Day 5, her cerebral spinal fluid cultures were negative, and her body temperature had returned to normal.
1. The surgical team performed Hannah’s skin prep without a complete head shave. How was this beneficial to the patient’s outcome?
a. The risk of permanent baldness is reduced when only small sections of the area are shaved.
b. Minimal shaving reduces the risk of skin abrasion created by clippers or razor.
c. Leaving hair around the incision permits prolonged contact of the antiseptic prep solution.
d. The remaining hair was used to retract the scalp for greater visibility.
2. How did the surgeon determine that the infection was not present at the time of the surgical procedure?
a. Baseline cultures were taken intraoperatively and were negative.
b. The surgical team performed hand surgical scrubs with chlorhexidine gluconate.
c. Antibiotics were not used during the procedure.
d. The causative microorganism is not native to the skin.
3. What was the most likely reason for Hannah’s infection?
a. The team used poor surgical technique.
b. The daily care staff’s hands were soiled.
c. The surgical team was not double-gloved.
d. The direct care staff had upper respiratory infections.
4. Why was iodophor the solution of choice for Hannah’s surgical prep?
a. Iodophor renders the skin sterile.
b. Her hair was freshly washed.
c. Iodophor is safe and effective for neuro preps.
d. Iodophor was more cost-effective to use.