Innovative Medical Products’ new Gel-Infused Memory Foam Pads are infused with proprietary formulated gel beads that provide 30 percent greater load distribution than standard memory foam pads, as demonstrated by independent pressure mapping tests. This greater load distribution reduces the possibility of pressure ulcers.
IMP’s Gel-Infused Memory Pads also provide for patient protection during surgical procedures, including robotic surgery. The new, larger pads are specially designed to accommodate most any size patient, meeting dimensional and density specifications for patient safety.
“As patients get bigger and bigger and actually begin to ‘spill over’ the edges of the boot and traditionally sized positioning pads,” noted Earl Cole, IMP vice president. “There comes a point where a standard memory foam pad will no longer be able to provide sufficient patient protection. Our gel-infused, larger pads are made to prevent just this from happening.”
IMP Gel-Infused Memory Pads come in a variety of shapes and sizes tailored to fit specific IMP patient positioning systems. These include:
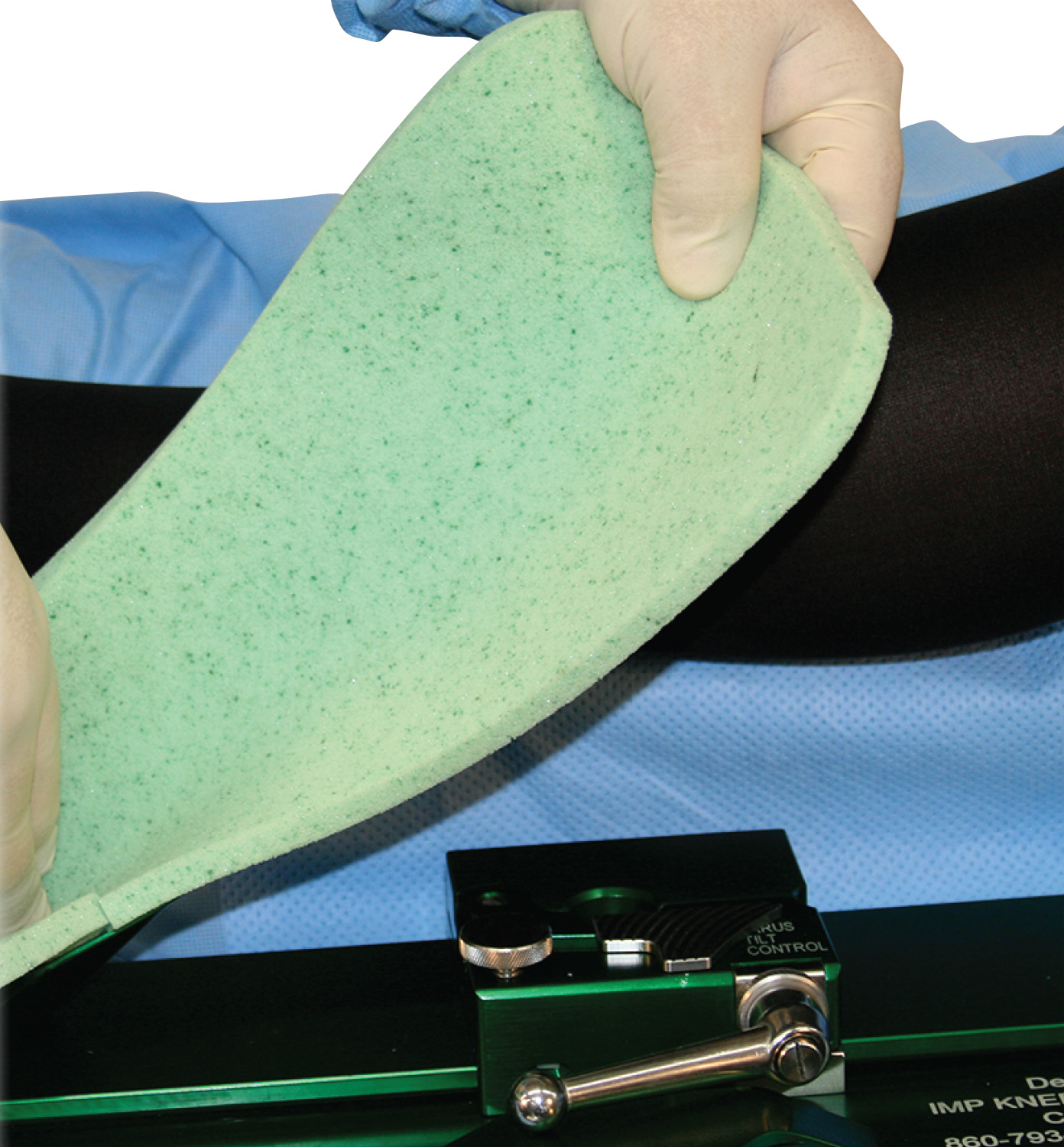
- Sterile Gel Foam Boot Pads designed for use with IMP’s entire family of De Mayo Knee Positioners; the boot pad is 1/2–inch larger than standard pads, improving coverage to the edges of the boot to accommodate larger patients.
- Sterile Gel-Infused Memory Foam Universal Distractor Pads that cover the entire edge of the boot; the patient’s foot in the boot is secured with IMP’s cohesive wrap that comes with the pad package.
- Gel-Infused Memory Foam Pad for IMP’s MorphBoard Positioning System, employing IMP’s modular pegboard lateral positioning method for total hip replacement surgery.