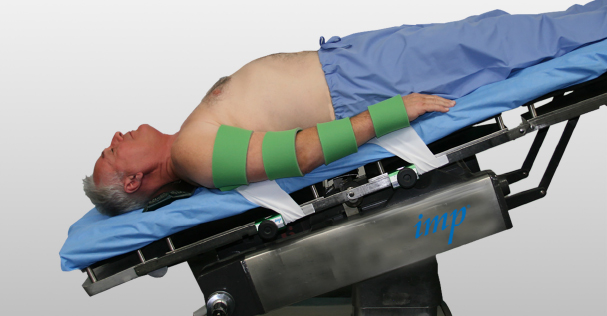
The Humbles TrenWrap™ positioning pad system securely anchors a patient wherever desired on the operating table prior to positioning in Trendelenburg for robotic and laparoscopic surgeries. The system’s Pre-Load™ Rail Clamps secure the straps of the positioning base pad, locking the pad tightly in position to the OR table’s side rails. The TrenWrap™’s proprietary Phase 4™ adhesive material applies directly to the patient’s skin, affixing the patient to the TrenWrap™ positioning pad system. In accordance with AORN guidelines, the TrenWrap™ system eliminates tucking sheets under the patient to hold the patient’s arms in place during surgery.