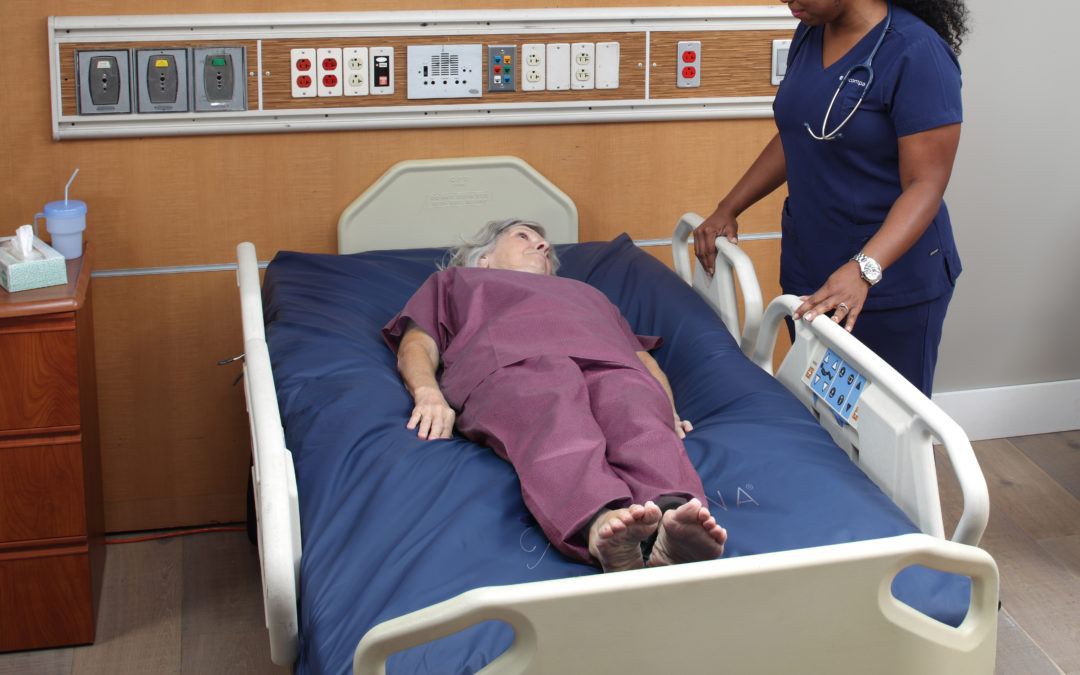
Encompass Group LLC premiered its Airisana Therapeutic Support Surface in June 2019 at the WOCNEXT 2019 Conference in Nashville, TN.
Over the last eighteen months, Airisana has been well received by the nursing community because it has provided a cost-effective solution that drives savings, supports a reduction in PI prevalence and risks, drives reductions in patient and staff injury and is simple to clean and store. Each unit comes with an intuitive soft touch user control panel that’s quiet enough to keep the healing environment comfortable, and still keeps control in the care provider’s hands.
Pressure injuries are generally not the primary reason a patient is admitted to a hospital, but they place a burden on patients and caregivers alike and can increase operational costs. “While many Hospital-Acquired Conditions (HACs) have decreased, pressure injuries remain a stubborn exception. Contributing factors such as increased acuities, decompensation, increased use of medical devices, and patient adaptation all play a role,” said Michelle Daniels, Airisana Managing Director for Product Development. “Airisana helps turn this tide around, providing ongoing support for both the patient and patient care team. It’s one more tool in their caregiving arsenal. Airisana has returned savings of more than 120% when purchased, compared to the cost of traditional, rented therapeutic support surfaces. This translates to a return on investment within 2-3 months. Combined with the benefits of its revolutionary design, and easy implementation, it helps nurses, clinicians and their institutions battle pressure injuries.”
Repeating pressure therapy patterns in traditional therapeutic support surfaces increases the probability that a patient’s body can adjust to the surface, negatively impacting healing. Airisana™ reduces this risk with randomized pressure therapy modes that promote sustained pressure reduction and combine the benefits of multiple pressure therapies into a constantly changing surface the body doesn’t get used to. These include: Alternating pressure, pressure redistribution, low air loss, and lateral rotation, immersion/envelopment. Airisana™ conforms evenly to irregularities such as a patient’s body contours. This puts the greatest amount of contact between skin and surface and ensures decreased pressure without interfering with patient mobility. And Airisana is even emergency-ready – with a CPR deflate function and a quick access hose that allows intervention when needed most. With turn assist reducing friction and shear risk, and preventing caregiver injury, nurses and clinicians can now easily reposition patients as needed.
More information is available at www.encompassgroup.com/airisana-reducing-pressure-injuries.