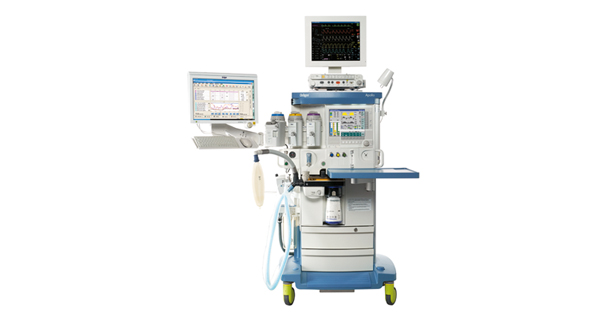
The Drager Apollo anesthesia system features a fully automatic self-test and an open-platform architecture. It can be customized with the Apollo Infinity patient monitoring system and peri-anesthesia information management system, as well as an optional web browser. The new Infinity Omega Solution uses a two-screen system approach so the clinician can see patient monitoring data and clinical data simultaneously. It includes an Infinity Delta patient monitor with the Infinity Docking Station and a 17-inch touch screen smart display.