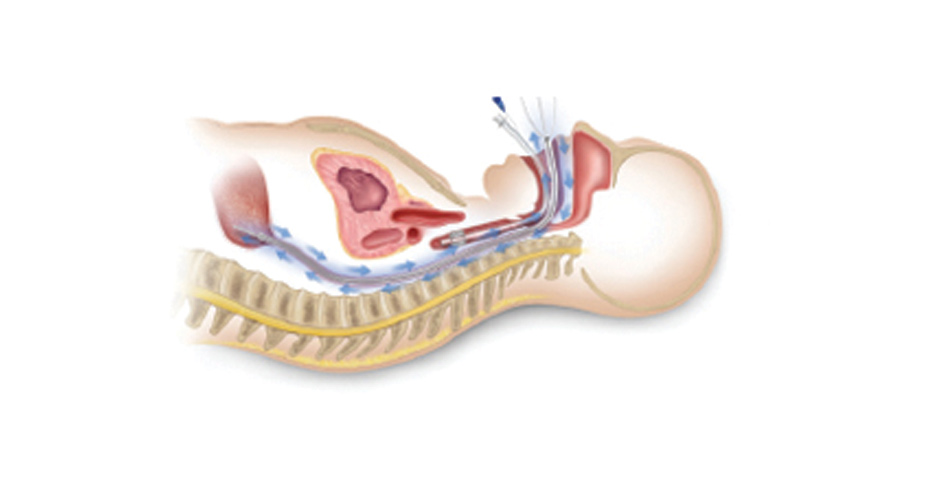
Advanced Cooling Therapy’s first product, the Esophageal Cooling Device (ECD), is showing benefits in the conduction of heat for warming of patients in a controlled manner in new clinical settings. A site in the European Union has used the ECD to keep challenging...
READ MORE