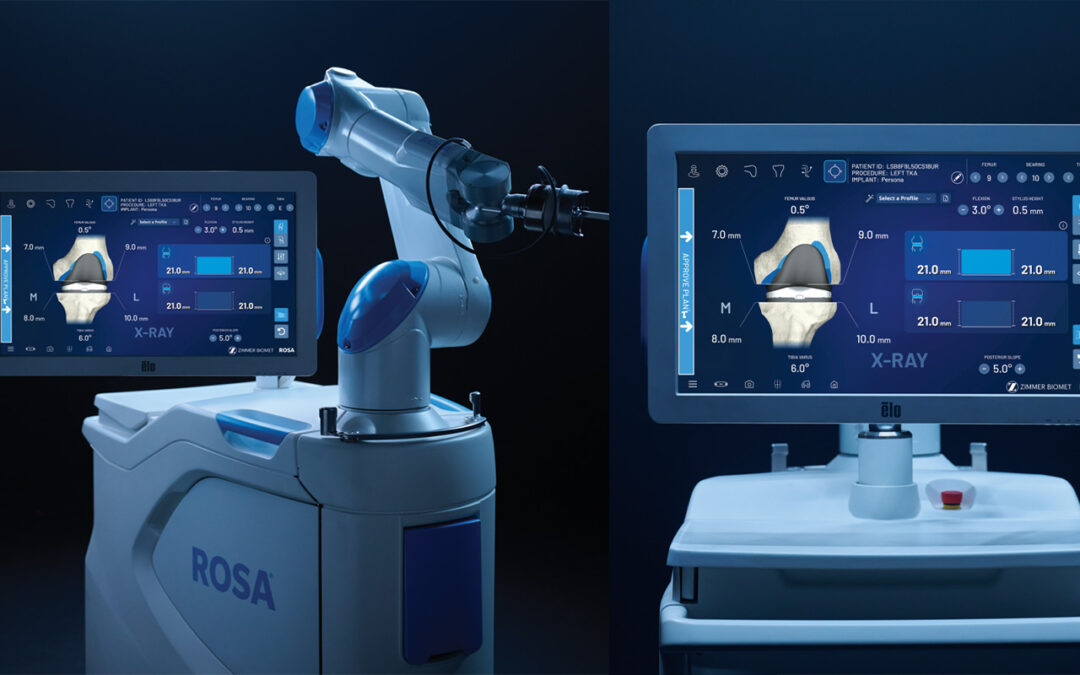
ROSA Knee with OptimiZe is a next generation robotic-assisted solution designed to support surgeon control, streamline workflow and enhance consistency in total knee arthroplasty. The system builds on the ROSA platform with intelligent planning tools, realtime tracking, and a simplified user interface that prioritizes efficiency and reproducibility.
READ MORE