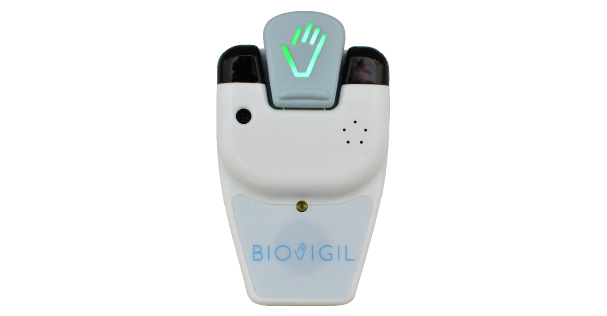
Biovigil is an automated hand hygiene compliance and awareness system that promotes safety at every patient interaction by intervening at the point of care to provide situation-specific hand hygiene reminders. Healthcare workers wear small, personal badges with on-board chemical sensors, so the system works with existing dispensers and sanitizers. After the badge validates hand hygiene, a green light is displayed, providing reassurance to patients and their families. Hospitals that use the BIOVIGIL system regularly achieve hand hygiene compliance rates well over 95 percent, compared to a national average of 45 percent.