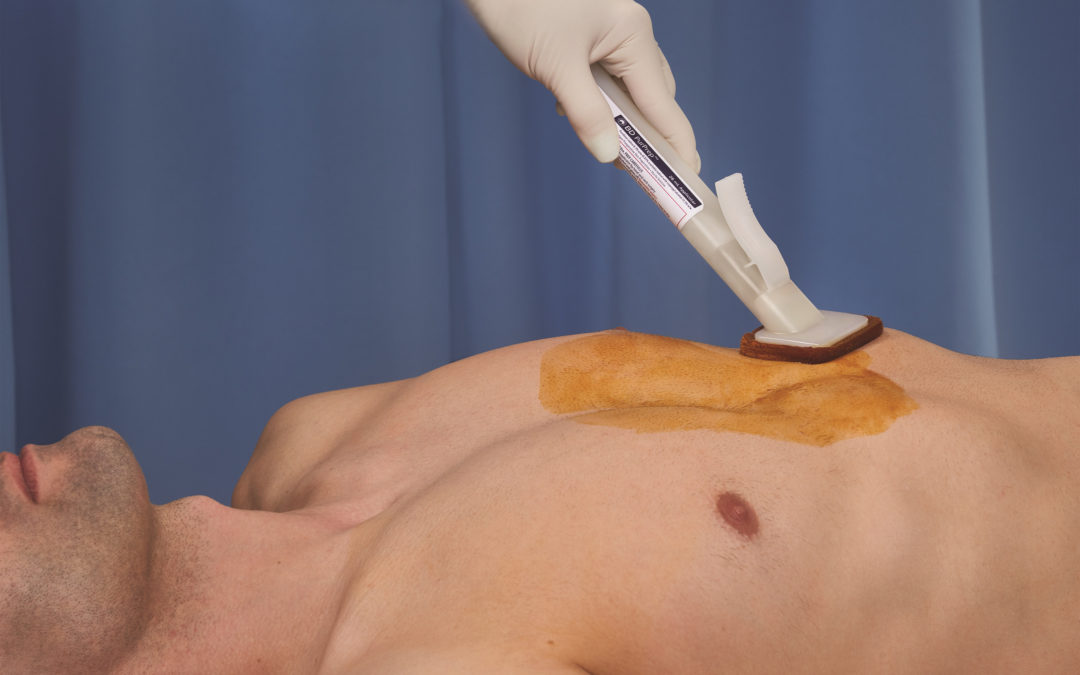
BD (Becton, Dickinson and Company) has announced the availability of BD PurPrep patient preoperative skin preparation with sterile solution, the first and only fully sterile povidone-iodine plus isopropyl alcohol single-use antiseptic skin preparation (PVP-I; 0.83% available iodine and 72.5% isopropyl alcohol) commercially available in the United States.
 BD PurPrep joins BD ChloraPrep patient preoperative skin preparation as the only fully sterile skin preparations available on the market that use a single-use applicator specifically designed to prevent cross-contamination and promote aseptic non-touch technique. The BD PurPrep patient preoperative skin preparation is an effective skin preparation alternative when the use of a chlorhexidine gluconate (CHG/IPA) is contraindicated or the patient is sensitive to CHG.
BD PurPrep joins BD ChloraPrep patient preoperative skin preparation as the only fully sterile skin preparations available on the market that use a single-use applicator specifically designed to prevent cross-contamination and promote aseptic non-touch technique. The BD PurPrep patient preoperative skin preparation is an effective skin preparation alternative when the use of a chlorhexidine gluconate (CHG/IPA) is contraindicated or the patient is sensitive to CHG.
“Though progress has been made, health care-associated infections remain a problem,” said Donald E. Fry, M.D., a nationally recognized expert in infection prevention. “Sterile single-use products have been shown to reduce the risk of outbreaks linked to microbial contamination of antiseptic products. By developing fully sterile PurPrep and ChloraPrep, BD is providing health care professionals with a more complete set of tools to reduce the risk of intrinsic contamination in antiseptic solutions.”
The new BD PurPrep formulation includes a fluid-resistant, film-forming polymer developed to facilitate drape adhesion and help create a durable antimicrobial barrier[1,2] that helps iodine bind to the skin for residual antimicrobial activity. In an irrigation study, mimicking real-world clinical practice, BD PurPrep patient preoperative skin preparation with sterile solution remained on the skin immediately following a saline challenge.[2]
“BD PurPrep strengthens our commitment to health care professionals and patients by providing an alternative fully sterile option to help reduce the risk of intrinsic contamination,” said Michael Cusack, BD, vice president/general manager of infection prevention. “By adding a non-CHG, fully sterile skin preparation option, BD is expanding the number of tools available to help reduce the risk of infection in patients with CHG sensitivities and during procedures when CHG/IPA is not indicated.”
BD PurPrep will replace Prevail and Prevail FX, and is available now through BD and other distribution partners.
Novel Sterilization Process
BD’s proprietary sterilization process was designed to achieve a minimum sterility assurance level (SAL) of 10-6, the same level required for injectable products.1 The SAL indicates there is less than one in 1 million chance that a sterile BD PurPrep or BD ChloraPrep applicator containing sterile solution will contain a single, viable microorganism following terminal sterilization of the ampoules through BD’s new sterilization process.1 While a product labeled as non-sterile does not suggest that it is contaminated with bacteria, it indicates that the contents have not been sterilized individually.
Like BD ChloraPrep, the new BD PurPrep sterile solution will include a mark to indicate that the solution is sterile. Though not required by the U.S. Food and Drug Administration (FDA), BD developed this mark to distinguish sterile solutions from non-sterile antiseptic skin preparations. In 2013, the FDA asked manufacturers to voluntarily change labeling on products (sterile vs. non-sterile) to further educate customers on the issue.
1 Jeng, D. A new, water-resistant, film-forming, 30-second, one-step application iodophor preoperative skin preparation. Am J Infect Control. 2001, 29(6):370-376
2 Mashiatulla, M. (2020). Iodine-based Pre-Operative Skin Preparation Product Performance on Porcine Abdominal Skin. Internal BD report: unpublished.