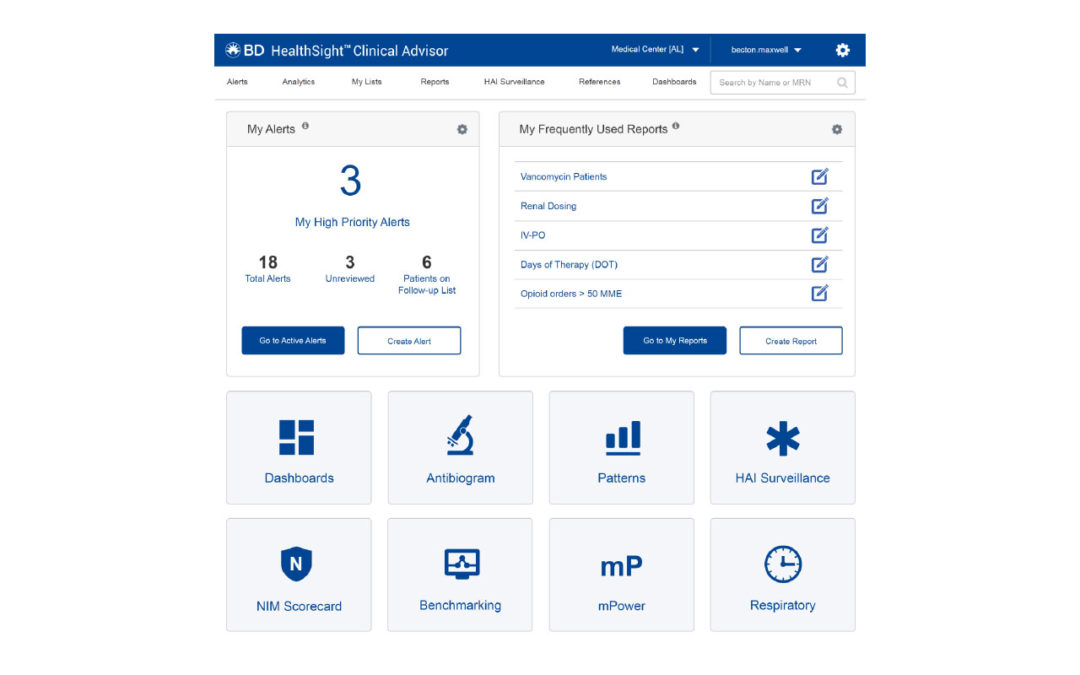
 BD HealthSight Clinical Advisor is a new functionality within the BD HealthSight connected medication management platform that aggregates disparate patient data to provide clinicians with the ability to receive near real-time medication stewardship alerts within the pharmacy workflow. These actionable alerts are delivered within the connected medication management process to help health care providers prevent not only the administration of inappropriate antibiotics, but also the potential waste of resources associated with compounding and preparing unnecessary medication for delivery. Clinical stewardship programs are also supported through customized dashboards, robust reports and the ability to automate the submission of antimicrobial utilization data for regulatory reporting.
BD HealthSight Clinical Advisor is a new functionality within the BD HealthSight connected medication management platform that aggregates disparate patient data to provide clinicians with the ability to receive near real-time medication stewardship alerts within the pharmacy workflow. These actionable alerts are delivered within the connected medication management process to help health care providers prevent not only the administration of inappropriate antibiotics, but also the potential waste of resources associated with compounding and preparing unnecessary medication for delivery. Clinical stewardship programs are also supported through customized dashboards, robust reports and the ability to automate the submission of antimicrobial utilization data for regulatory reporting.