A new study suggests that some hospitals may classify admissions in a way that exempts them from elective-based PSI scores, a practice that may lead to less reliable PSIs.
READ MORE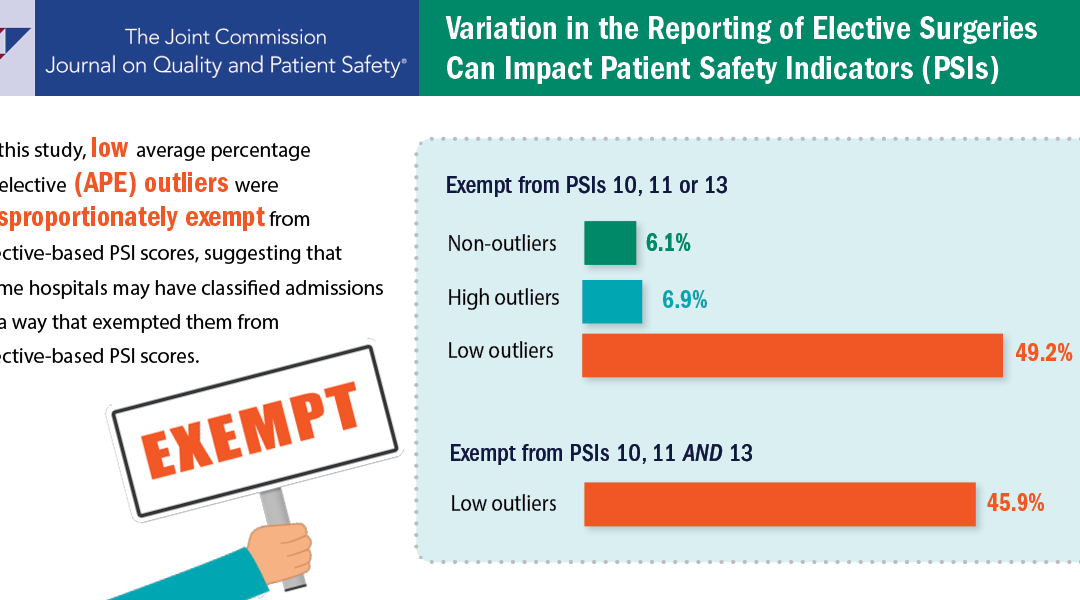
Articles Posted by

by OR Today Magazine | Aug 3, 2022
A new study suggests that some hospitals may classify admissions in a way that exempts them from elective-based PSI scores, a practice that may lead to less reliable PSIs.
READ MORE
by OR Today Magazine | Aug 2, 2022
Today during the virtual Achieving Accreditation conference, the Accreditation Association for Ambulatory Health Care (AAAHC) revealed the winners of the Bernard A. Kershner Innovations in Quality Improvement (QI) Award, shining a spotlight on health care...
READ MORE
by OR Today Magazine | Aug 1, 2022
Having issues viewing the content embedded below? Click here for a direct link....
READ MORE
by OR Today Magazine | Aug 1, 2022
Following a two-year, pandemic-related hiatus of the Healthcare Sterile Processing Association (HSPA) in-person annual conference, this year’s event brought more than 900 attendees together for five days of top-quality education and networking in San Antonio, Texas. The expansive and technologically advanced Henry B. Gonzales Convention Center housed the conference and expo. It provided an ample and inviting space for attendees to learn, grow, share best practices, and engage with peers, renowned industry experts and leading product and service vendors in the sterile processing (SP) space.
READ MORE
by OR Today Magazine | Aug 1, 2022
In addition to instruments in disrepair making their way to the sterile field, Joint Commission surveyors have unfortunately noted with increasing frequency, the reprocessing of single-use devices (SUDs) – items labeled by their manufacturer as “single use” or disposable. These devices often consist of lower-quality material and as a result, develop pitting and oxidation which indicates possible hazard to a surveyor. Reprocessors are regulated by the Food and Drug Administration (FDA) and are subject to all regulatory requirements currently applicable to the original device manufacturer, including premarket submission requirements. If a health care organization reprocesses a single-use device, it is dangerously allowing itself to become a manufacturer of a medical device – and thus assumes all of the manufacturer’s liability, should that device fail. In addition, it takes on the liability of possible FDA rule violation. Single-use instruments must be removed from trays and discarded or sent to an FDA-approved reprocessor of SUDs.
READ MORE
by OR Today Magazine | Aug 1, 2022
The Tru-D device, part of PDI’s infection prevention solutions, is a portable UVC disinfection robot that delivers one automated, measured dose of UVC to consistently disinfect a room, resulting in the ability to document disinfection results after each and every room...
READ MORE

For over 20 years, OR Today has provided perioperative and SPD professionals with up-to-date news and information about their profession. The magazine aims to educate readers about new guidelines, techniques, and equipment, as well as practical information for career building, problem-solving and overall well-being.
© 2026 MD PUBLISHING