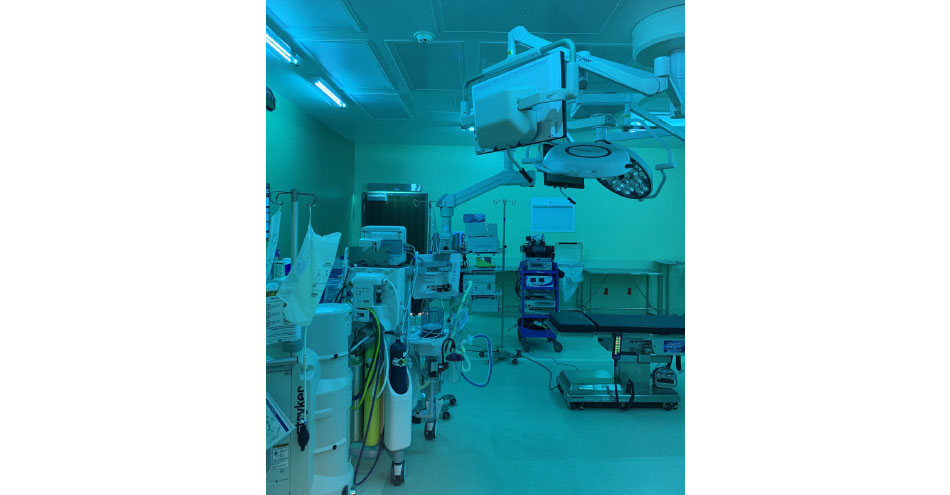
The UVC OR package is the only whole room disinfection solution designed to be used before, during and after a procedure. An automatic daily disinfection cycle requires no staff to initiate and all of the data is collected and saved. The system is unique as it delivers the energy from the ceiling down and this helps to eliminate shadows so one cycle treats the entire room. The package requires no additional staff so facilities save on labor hours and additional FTEs. The package is designed for the space to maximize effectiveness if it is glowing blue it is being disinfected.