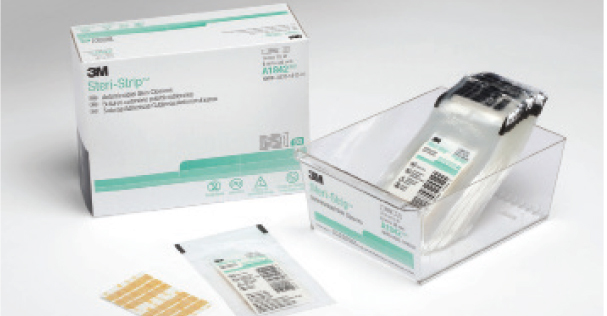
The 3M™ Steri-Strip™ Antimicrobial Skin Closure is a small but effective solution for reducing the risk of surgical site infections (SSIs), the second most common hospital-acquired infection. The adhesive skin closure product has broad-spectrum activity against the pathogens most commonly causing SSIs and it is the only adhesive skin closure product on the market with antimicrobial properties. Numerous clinical studies have shown that wounds treated with Steri-Strip™ Skin Closures exhibit lower infection rates than those closed with invasive sutures or staples. Studies have also shown better cosmetic results than sutures or staples.