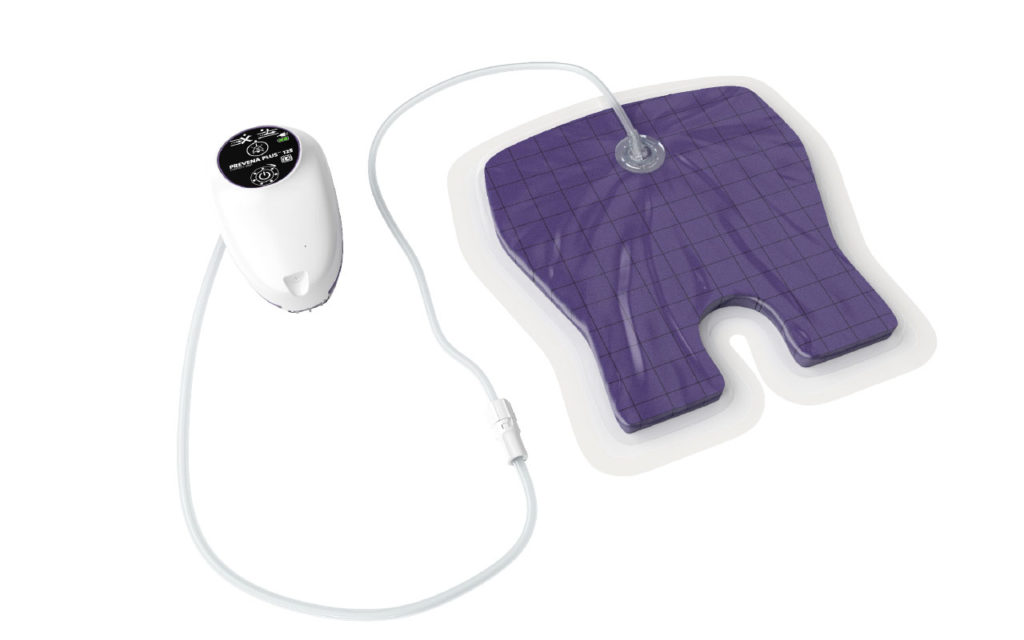
The PREVENA RESTOR AXIO•FORM Incision Management System delivers negative pressure wound therapy designed to manage post-operative incisions, as well as the surrounding soft tissue envelope. PREVENA RESTOR AXIO•FORM System helps stabilize the incision and surrounding soft tissue, reduce edema, and helps enhance post-operative recovery. This is the third offering in the PREVENA RESTOR Therapy portfolio, launched in 2019, to optimize post-surgical care and expand the company’s specialty surgical offerings. Please refer to the PREVENA RESTOR AXIO•FORM System Instructions For Use for important safety information.
NOTE: Specific indications, limitations, contraindications, warnings, precautions and safety information exist for these products and therapies. Please consult a clinician and product instructions for use prior to application. Rx only.









