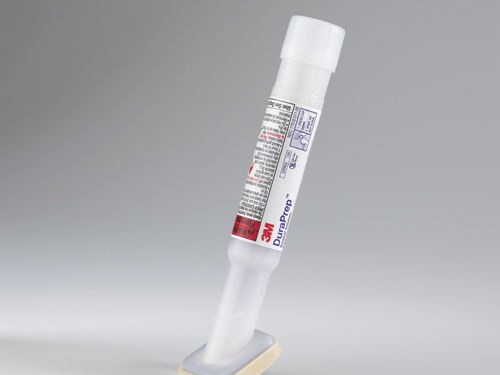
DuraPrep meets AORN and CDC guidelines for the reduction of surgical site infections, as well as the FDA tentative final monograph (TFM) log reduction requirements. Its active ingredient is iodine povacrylex. DuraPrep solution is persistent at least 48 hours against resident bacteria after a blood and saline (fluid) challenge, the company says. DuraPrep is designed to be applied in a single, painted coat with no scrubbing required. A 26-millileter applicator of Dura- Prep covers 15 inches by 30 inches, or 450 square inches, according to labeling.