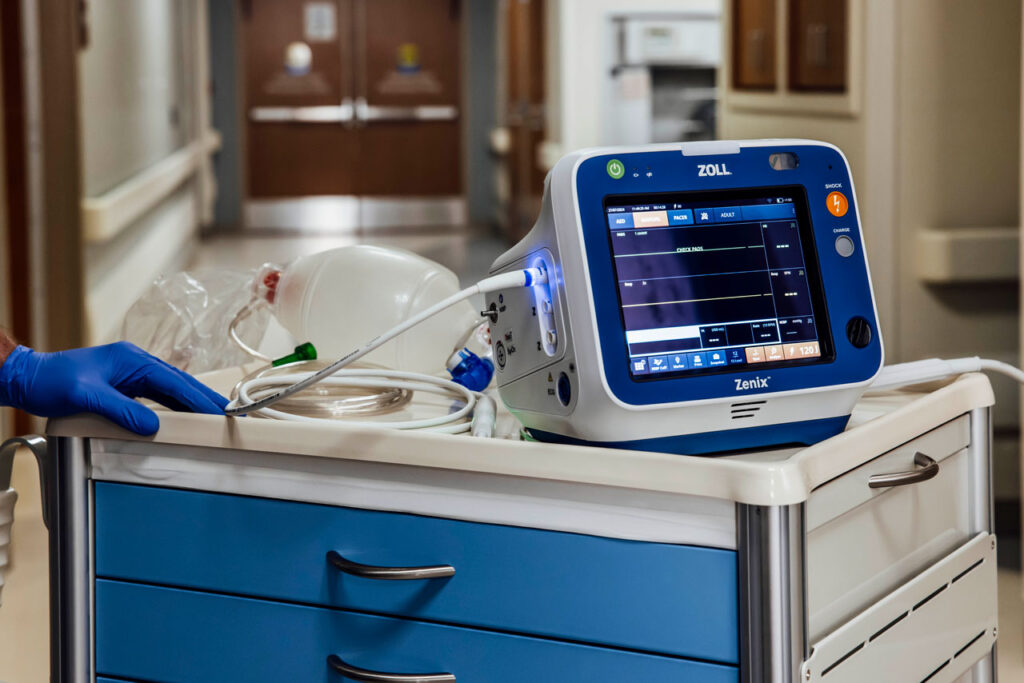
ZOLL, an Asahi Kasei company that manufactures medical devices and related software solutions, has announced that Zenix, the company’s most clinically advanced and easy-to-use professional monitor/defibrillator, has received premarket approval (PMA) from the U.S. Food and Drug Administration (FDA).
The Zenix monitor/defibrillator is a groundbreaking device that redefines efficiency, clarity and intelligence in EMS and hospital settings. Built from years of feedback from customers, Zenix combines intuitive design with powerful functionality to enhance patient care and automate workflows for ease-of-use. Featuring a large, durable touchscreen, Zenix provides critical information when it’s needed. With on-the-fly customization, healthcare professionals can make real-time adjustments, helping them stay in control during high-pressure situations.
Equipped with ZOLL’s innovative Real BVM Help and exclusive Real CPR Help technology, Zenix gives healthcare providers real-time clinical feedback to improve ventilation quality and deliver high-quality CPR, according to a press release. The advanced technology of Zenix empowers EMS teams and hospital clinicians to make informed, confident decisions and ensure they are delivering exceptional patient care, every time.
“Whether you are a hospital clinician or an EMS professional, the ZOLL Zenix monitor/defibrillator is designed to work the way you work,” said Elijah White, president of ZOLL Acute Care Technology. “Zenix is our most advanced monitor/defibrillator, and yet it’s incredibly easy to use – virtually all functions of the device are accessible with three screen-touches or less. Along with the X Series and R Series, Zenix extends ZOLL’s leadership in professional monitors/defibrillators with unique features and intuitive design.”
The Zenix monitor/defibrillator is available now to customers in the U.S. and Canada. For more information, visit zoll.com/zenix.










