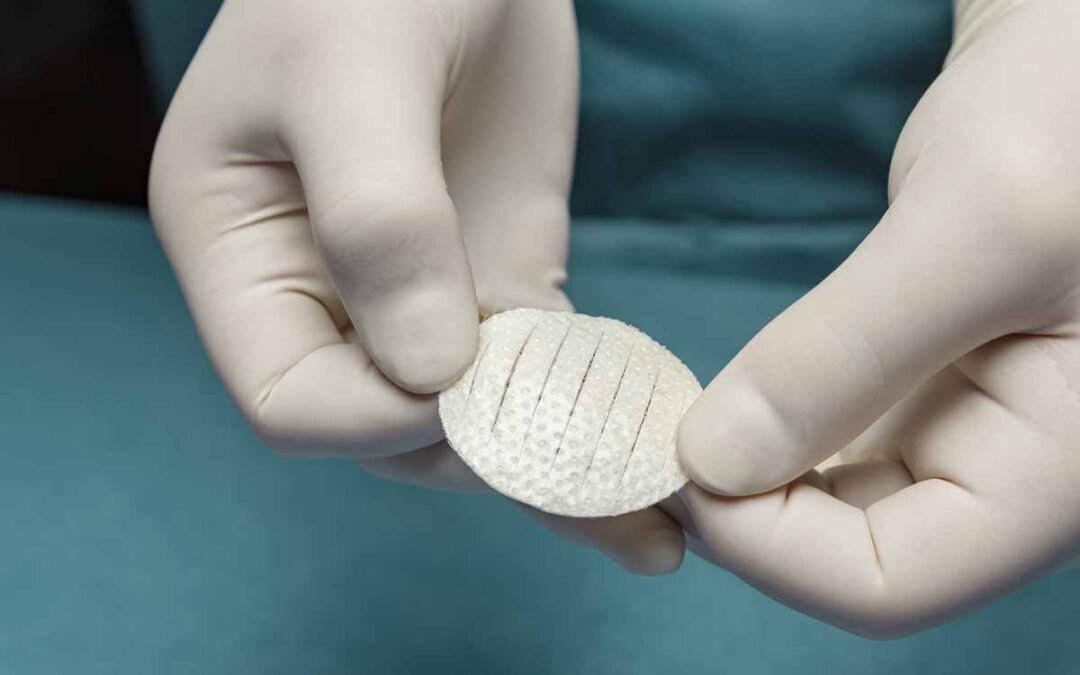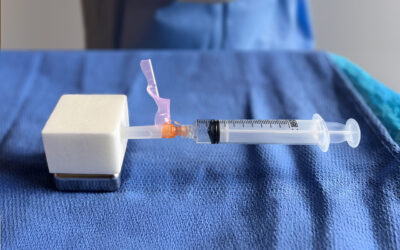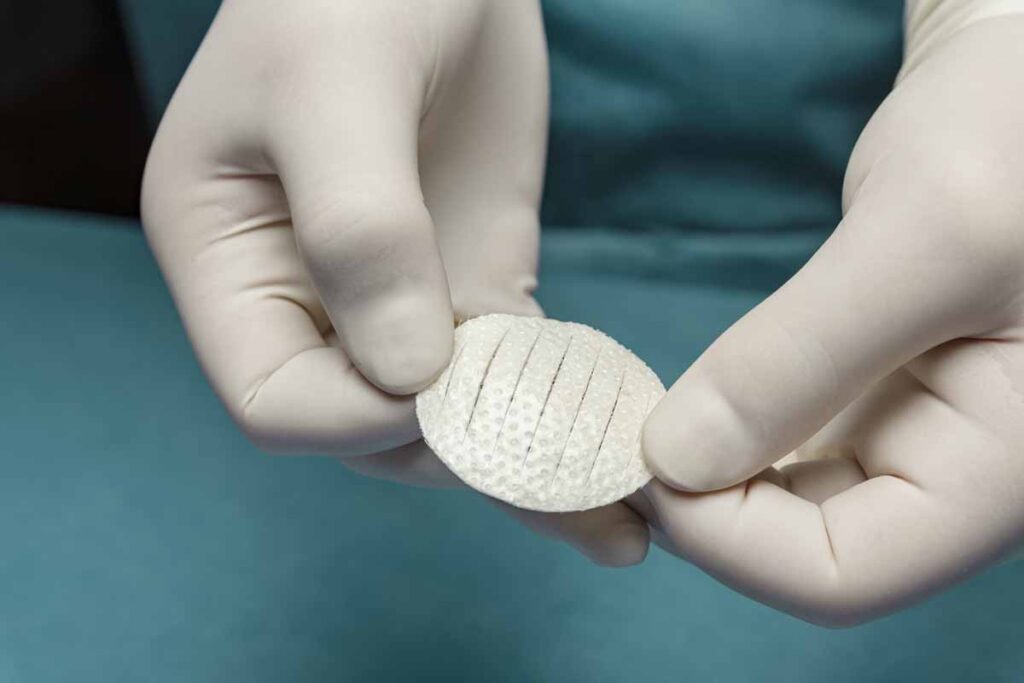
Kerecis, the company pioneering the use of sustainably sourced fish skin and fatty acids in cellular therapy and tissue regeneration and protection, recently announced the availability of Shield Standard, which combines a fish-skin graft and silicone backing for efficient treatment of acute and chronic wounds. Shield Standard is a part of the Kerecis Shield product family, which is designed for managing chronic and acute wounds such as diabetic foot ulcers, pressure ulcers, vascular ulcers, post-Moh’s surgical wounds, and draining wounds. The grafts contain essential natural skin elements and are fenestrated to allow adequate wound drainage for appropriate wound moisture at the wound bed. Kerecis develops products from fish skin and fatty acids for cellular therapy, tissue regeneration and protection. When grafted onto damaged human tissue or implanted, the patented material supports the body’s own processes to heal and regenerate. Because no disease-transfer risk exists between cold-water fish and humans, the Kerecis fish skin is only gently processed and retains its similarity to human tissue. The gentle processing preserves the skin’s original three-dimensional structure, maintaining its inherent natural strength, complexity, and molecules (such as fatty acids). Clinical studies have found that the Kerecis products heal wounds faster than competing products and are cost effective. Kerecis is the only approved manufacturer of medical devices containing intact fish skin in the world.